Rectal Cancer MR staging and restaging- update 2026
Doenja Lambregts¹², Monique Maas¹², Max Lahaye¹², Regina Beets-Tan² and Robin Smithuis³
¹Radiology Departement of the Netherlands Cancer Institute in Amsterdam, ²Maastricht University (GROW) and ³Alrijne Hospital in Leiden, the Netherlands
Publicationdate
The rectal cancer treatment landscape is rapidly evolving.
Key historical advances - standardized total mesorectal excision (TME), neoadjuvant (chemo)radiotherapy for high-risk tumors, and MRI-based risk stratification - have significantly reduced local recurrence rates.
Recent developments, including organ-preserving strategies (watch-and-wait) and total neoadjuvant treatment (TNT), have further increased the need for precise radiological staging, response monitoring, and detection of regrowth or recurrence.
MRI remains the gold standard for local staging and restaging in rectal cancer, playing a pivotal role in guiding treatment decisions.
Updates in this edition:
- Treatment Overview: Current rectal cancer therapies and their impact on radiological staging.
- MRI Interpretation: Key pearls and pitfalls in reading and reporting rectal cancer MRI.
- Reporting Templates: Updated templates for primary staging and restaging, aligned with the 2026 ESGAR rectal imaging guidelines [ref1] [ref2]
- Renewed guidelines for assessment of lymph nodes and tumor deposits.
- A stepwise approach for restaging to guide the choice between surgery and organ-preservation.
Acknowledgments: The authors thank Rhiannon van Loenhout and Frank Zijta for their contributions to earlier versions.
Introduction
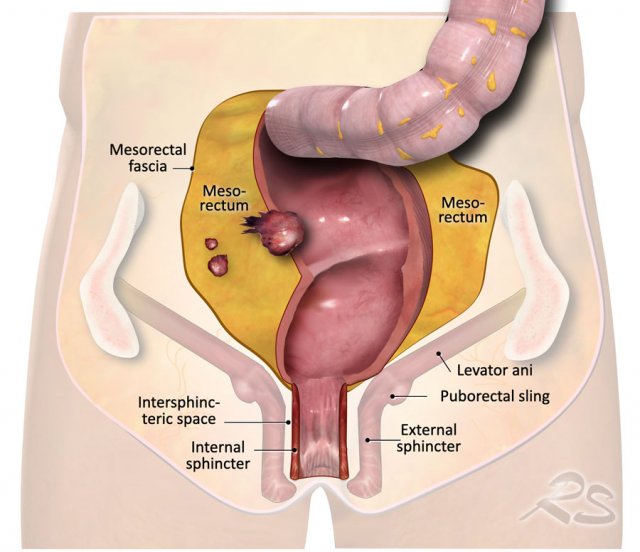 The illustration shows the mesorectum and the mesorectal fascia, which is the plane for TME resection and the relation of the rectum to the anal sphincter and pelvic floor.
The illustration shows the mesorectum and the mesorectal fascia, which is the plane for TME resection and the relation of the rectum to the anal sphincter and pelvic floor.
Evolution of Rectal Cancer Management
Over recent decades, rectal cancer treatment has shifted from a surgery-centric approach to a multimodal, imaging-guided strategy.
Total mesorectal excision (TME)—en bloc removal of the mesorectal compartment along the mesorectal fascia (MRF)—remains the surgical cornerstone.
The introduction of neoadjuvant radiotherapy and chemoradiotherapy (CRT) for high-risk tumors has markedly reduced local recurrence, with many patients achieving a complete response.
This has facilitated organ-preserving strategies, such as ‘watch-and-wait’ for clinical complete responders, now endorsed in clinical guidelines. CRT is increasingly used even in low-risk tumors to enable organ preservation.
Recent advances include total neoadjuvant treatment (TNT), combining systemic chemotherapy with (chemo)radiotherapy, which improves disease-free survival and complete response rates, particularly for very high-risk tumors or patients prioritizing organ preservation.
Additional innovations include immunotherapy for selected subgroups, and refined local excision and radiotherapy techniques for early-stage, superficial tumors, or small residuals post-neoadjuvant therapy.
MRI for Risk Stratification
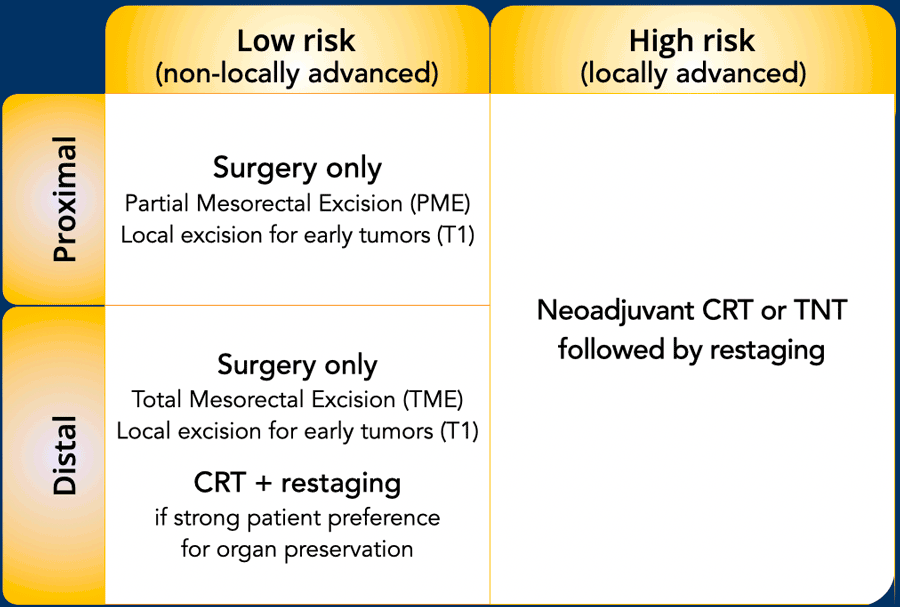
MRI local staging stratifies patients by risk and guides treatment selection.
Risk criteria vary widely across trials, countries, and guidelines, and continue to evolve.
Treatment decisions are increasingly influenced by patient preferences, with an enhanced focus on organ preservation and quality of life.
A global overview of current approaches is presented in the Figure, though local and national protocols may differ.
Most guidelines classify rectal cancers as either non-locally advanced (low risk) or locally advanced (high risk) [ref1] [ref2].
Low-Risk / Non-Locally Advanced Rectal Cancer
Low-risk features—though definitions vary by guideline—typically include:
- T1–T2 tumors
- No EMVI (extramural vascular/venous invasion), no mesorectal fascia (MRF) involvement.
- Management: Typically surgery alone; neoadjuvant treatment generally not required (unless organ preservation is the goal). Early stage T1 tumors may be eligible for local excision, following endoscopic/endorectal ultrasound confirmation.
Note: Guidelines vary on whether T3 (or more specifically T3cd) disease alone constitutes high risk. Current guidelines also de-emphasize lymph node status; suspected mesorectal lymph node metastases (particularly if few) are not necessarily classified as high risk.
High-Risk / Locally Advanced Rectal Cancer
High-risk features—though definitions vary by guideline—typically include:
- T4 disease (some guidelines also include T3 or advanced T3cd)
- EMVI
- Advanced nodal disease (especially N2 or lateral lymph node involvement)
- Tumor deposits
- Mesorectal fascia (MRF) involvement
- Management: Patients with one or more high-risk features usually receive long-course chemoradiotherapy or total neoadjuvant treatment to achieve tumor downsizing/downstaging, facilitate R0 resection, and potentially enable organ preservation.
In the current Dutch guidelines, locally advanced disease is defined by the presence of cT4b, MRF+, lateral lymph node metastases, EMVI+ and/or tumor deposits.
These guidelines further emphasize that cT4a and mesorectal N+ disease should not be considered locally advanced in the absence of other high-risk features [ref].
MRI in Neoadjuvant Treatment Response Assessment
In patients receiving neoadjuvant therapy, MRI—together with endoscopy—is pivotal for evaluating treatment response. It distinguishes between:
- Poor responders requiring radical surgery
- Potential candidates for organ preservation
Restaging Timing:
- Recommendations on when to perform restaging MRI vary among guidelines from 6–12 weeks post-treatment.
- The Dutch guidelines specify 6–8 weeks after radiotherapy-based neoadjuvant treatment for neoadjuvant chemotherapy, restaging is advised 3 weeks after the final cycle [ref].
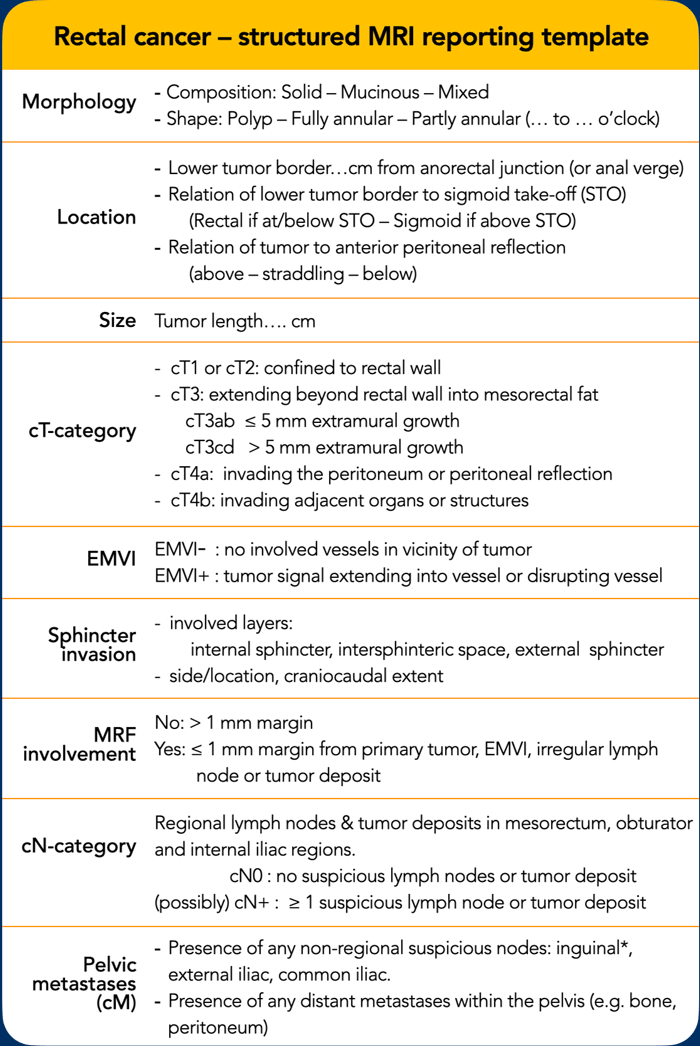
Structured Reporting Checklist
A comprehensive MRI report must include:
- All risk factors used for treatment stratification
- Precise tumor location and its relationship to adjacent anatomical structures, to guide surgical planning
This template adheres to the 2026 ESGAR structured reporting guidelines [ref] and aligns with the latest Dutch national recommendations [ref].
cT-category
cT4b: invading adjacent organs or structures includes:
- Pelvic floor/sidewall muscles
- External sphincter muscles
- Vessels, nerves, bones,
- Other bowel loops and anatomic compartment or fat outside MRF
cN-category
Highlight in report the level of most proximal N+ node (to guide radiotherapy and surgical planning, presence of lateral (obturator, internal iliac) N+ nodes, incl. size and location and presence of tumor deposits.
* Note: Inguinal nodes are considered cN (and not cM) in distal tumors extending below dentate line
TNM-prefixes
“c” is used to indicate the clinical stage, determined before treatment.
When defined based on imaging, the prefix “i" (imaging) or “mr” (MRI) are sometimes used as alternatives.
“y” is used to restage tumors after neoadjuvant treatment (chemo and/or radiotherapy) and can be used for both clinical staging (ycTNM) as well as pathological staging (ypTNM).
“p” indicates the final TNM stage as determined at histopathology after surgery.
In the following chapters we will discuss the various items listed in the reporting template in further detail and provide tips for image interpretation.
Morphology
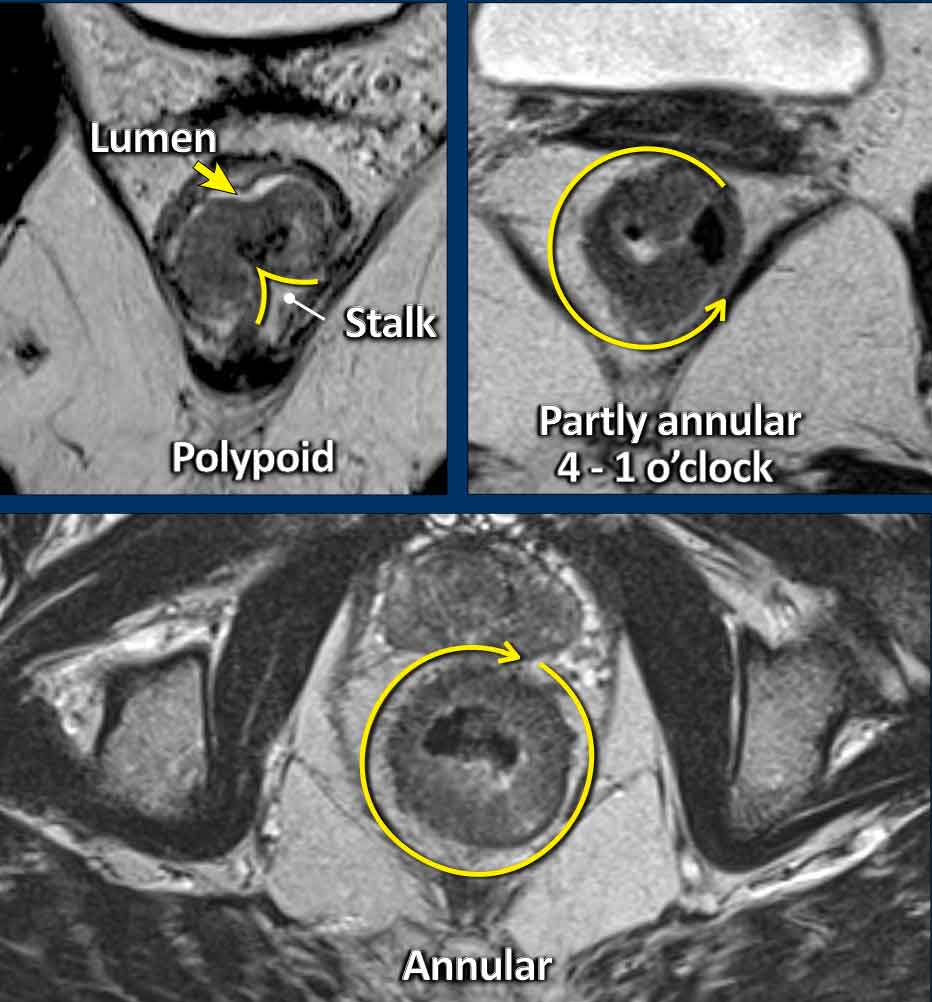
Polypoid and Sessile tumors
Rectal adenocarcinomas usually originate from adenomas, which may be:
- Polypoid: Raised on a stalk or focal attachment, often low-grade (T1–T2), projecting into the lumen.
- Sessile: Flat, broad-based, typically presenting as partial or complete annular wall thickening.
Invasive Margin: The tumor’s attachment site to the rectal wall—the invasive margin—is the site where the tumor may invade beyond the rectal wall, and is therefore critical for T-staging and assessing extramural extension.
Tumor Circumference: Describe the degree of wall attachment in the report using either:
- Clock-face notation (e.g., “from 3 to 7 o’clock”)
- Prose (e.g., “left anterolateral”).
Solid vs. Mucinous Rectal Adenocarcinomas
Clinical Relevance
Mucinous adenocarcinomas carry a poorer prognosis and typically exhibit a suboptimal response to neoadjuvant therapy compared to solid tumors.
MRI Characteristics:
- Mucinous tumors: High (bright) signal on T2-weighted MRI
- Solid tumors: Intermediate signal
- Mixed tumors: Both solid and mucinous components are common within the mucinous subgroup
Reporting Note: Due to the high risk of biopsy sampling error (especially in mixed tumors), radiologists should explicitly document the presence of mucin on MRI.
Signet-Ring Cell Rectal Adenocarcinoma
Epidemiology & Prognosis
A rare mucinous subtype (±1%), signet-ring cell carcinoma is associated with high rates of nodal/distant metastasis and poor overall survival.
MRI Features
- Often challenging to identify
- Typically presents as long-segment, diffuse bowel wall thickening
- Submucosal growth pattern, yielding a "target" appearance on axial images
- Diffuse mesorectal fat infiltration is common
Images
The provided images demonstrate a signet-ring cell carcinoma with diffuse rectal wall thickening, the classic target appearance, and mesorectal fat infiltration.
Location
Differentiating Rectal from Sigmoid Cancer
Accurate discrimination between rectal and sigmoid cancer is critical, as their management strategies differ significantly:
- Sigmoid cancer: Standard treatment is upfront surgical resection.
- Rectal cancer: Treatment is risk-stratified, ranging from surgery alone (low-risk) to long-course neoadjuvant chemoradiotherapy (high-risk). Surgical approaches also vary between the two entities.
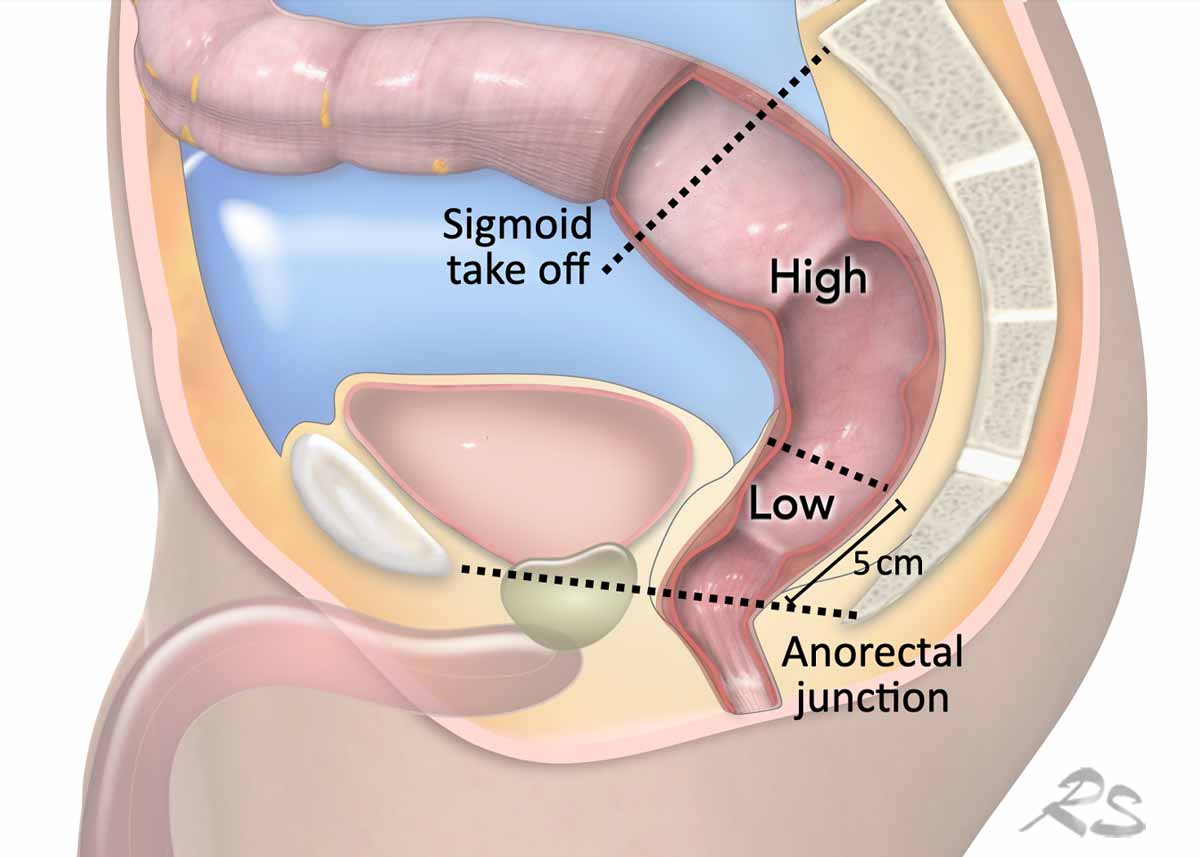
Sigmoid take-off
The sigmoid take-off (STO) as an anatomical landmark was established in 2019 by an international consensus panel as the preferred imaging landmark for distinguishing the rectum from the sigmoid colon (ref).
This definition has been adopted by ESGAR and Dutch guidelines.
Rectal Tumor location
- Low: Lower tumor border ≤5 cm from the anorectal junction
- High: Lower tumor border >5 cm from the anorectal junction, up to the sigmoid take-off
Identifying the Sigmoid Take-Off on MRI
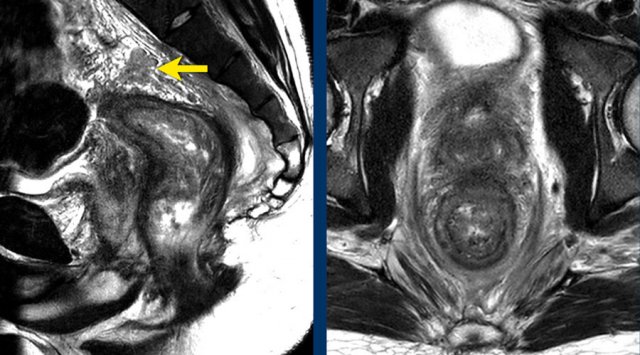
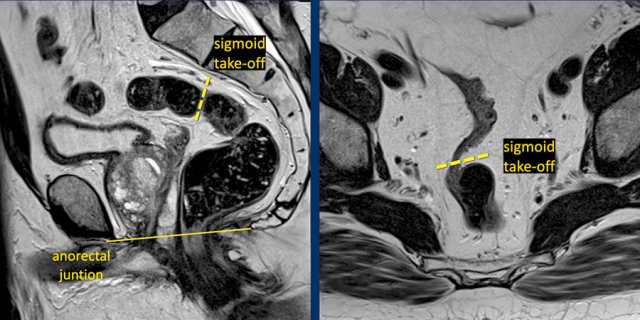
The sigmoid take-off (STO) is best recognized on:
- Sagittal MRI: The point where the sigmoid colon sweeps horizontally away from the sacrum
- Axial MRI: The point where the sigmoid projects ventrally (see Figure)
While anatomical variations or sequence angulation may occasionally complicate identification, the STO remains an intuitive and reliable landmark.
Tumors with a lower border proximal to the STO (above the rectosigmoid junction) are classified as sigmoid tumors.
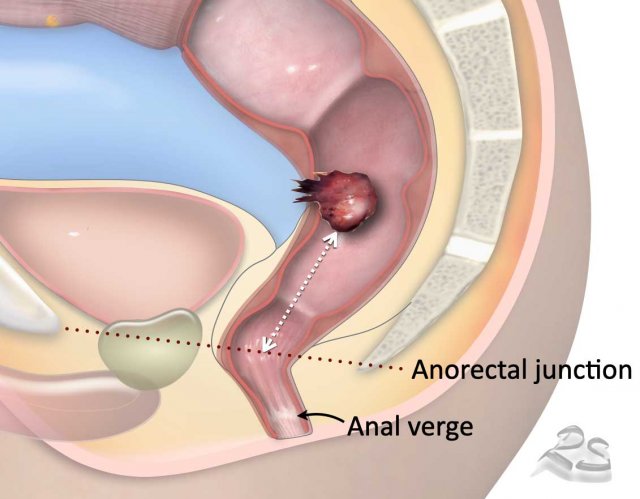
Tumor Height Measurement
Anorectal junction (most commonly used landmark):
- Defines the transition between the anal canal and distal rectum.
- Located at the anorectal angle, formed by puborectalis muscle contraction.
- On sagittal MRI, typically aligned with an imaginary line between the lower margins of the sacrum and pubic bone.
Anal verge (alternative landmark):
- Transition between anal epithelium and perianal skin
- Typically well visualized on sagittal MRI
Tumor Height Assessment:
- Standard method: Measure from the lower tumor border to the anorectal junction (or anal verge).
- Alternative: Measure from the anal verge (junction of anal epithelium and perianal skin).
- ESGAR recommendation: Use one or more straight lines along the central rectal lumen for consistent measurement [ref].
- In some countries, tumor height is referenced to the anterior peritoneal reflection.
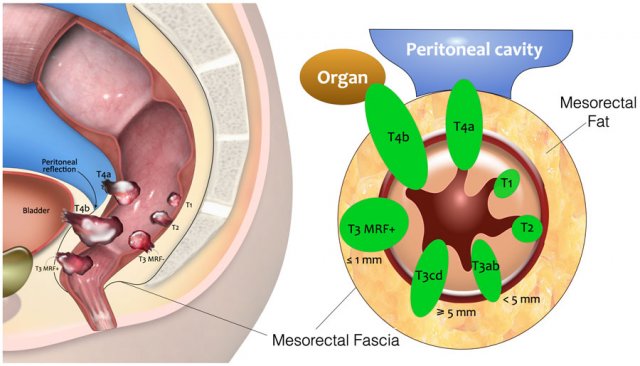
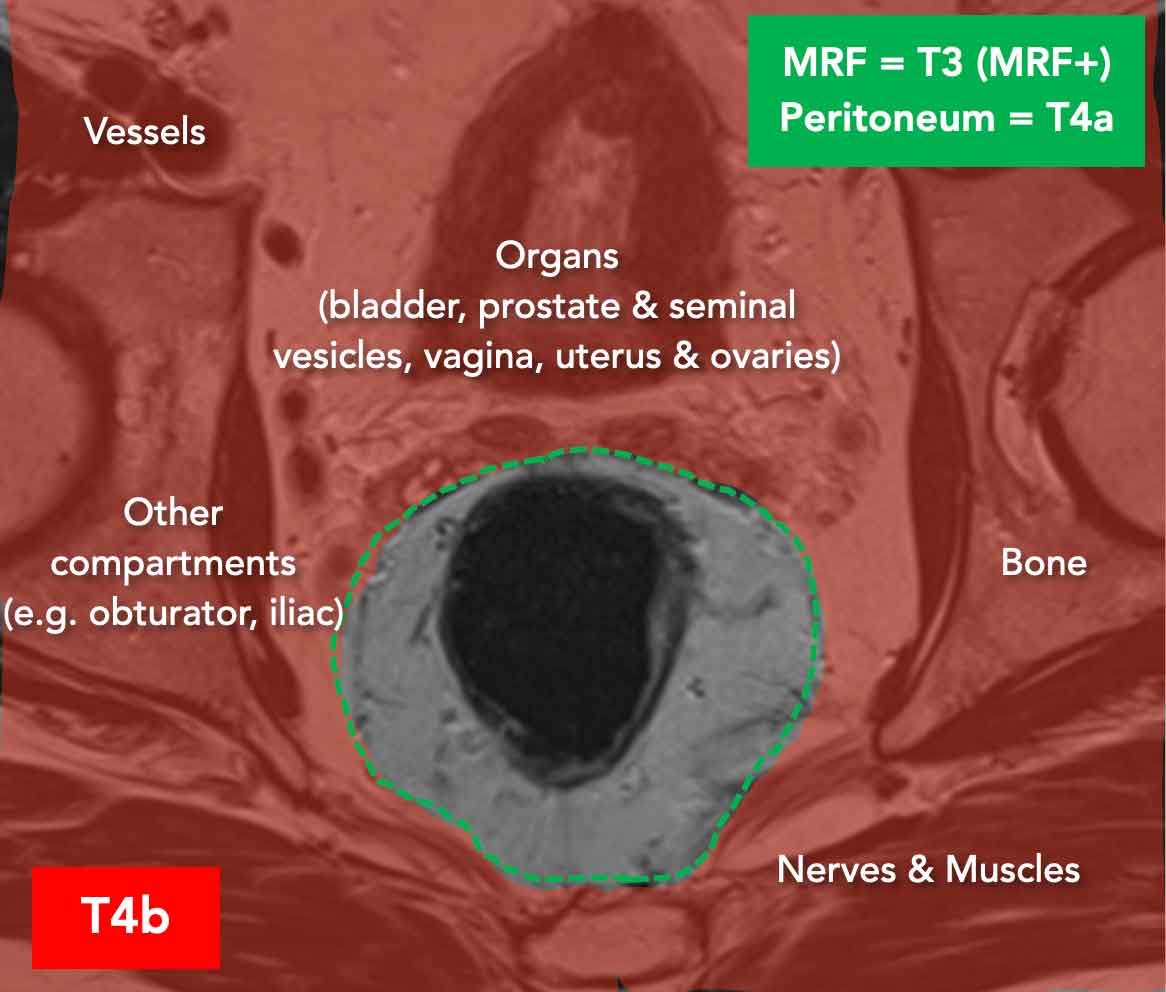
T-category
T-Categorization of Rectal Tumors: Sagittal and Axial Illustration.
- T1: Invasion into the submucosa
- T2: Invasion into the muscularis propria (outer muscular layer of the rectal wall)
- T3: Extension into the mesorectal fat
- T3 MRF–: Does not reach the mesorectal fascia (MRF)
- T3 MRF+: Invades or lies ≤1 mm from the MRF
- T4a: Invasion of the peritoneum/peritoneal reflection
- T4b: Invasion of adjacent organs/structures outside the mesorectum
The table shows an overview of the T-staging.
T1/T2 Confined to the rectal wall; often indistinguishable on MRI
T3 Extends into perirectal fat:
- Early T3 (T3ab): Limited extension (<5 mm)
- Advanced T3 (T3cd): Extensive invasion (>5 mm)
T4 | Extends beyond the mesorectum:
- T4a: Peritoneal/peritoneal reflection invasion
- T4b: Adjacent organ/structure invasion |
T4b Definition
The TNM system lacks a precise definition of "structures."
In 2021, an international expert panel (radiologists, surgeons, radiation oncologists, pathologists) proposed specific structures for T4b classification [ref], now adopted by ESGAR and Dutch guidelines (see Table).
T1–T2 Tumors
T1–T2 tumors are confined to the bowel wall and carry a relatively favorable prognosis.
- T1 tumors
- Limited to the submucosa
- Depth of submucosal invasion (Sm1 = superficial one-third; Sm2 = middle third; Sm3 = deep third, approaching muscularis) determines lymph node metastasis risk and local excision feasibility
- T2 tumors
- Invade the muscularis propria but do not extend beyond it
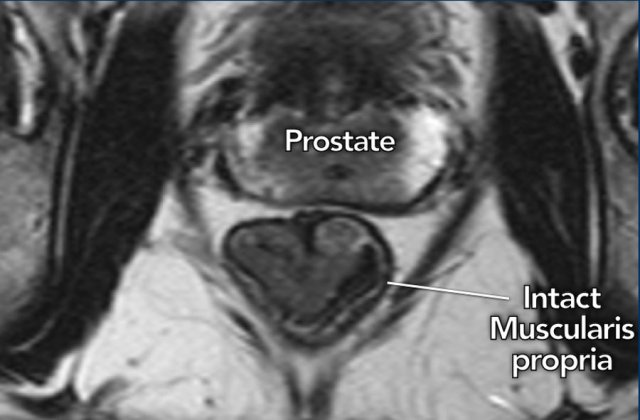
Key MRI Finding:
- Intact muscularis propria (hypointense line surrounding the rectum) confirms T1–T2 tumor
- All three planes (axial, sagittal, coronal) must demonstrate the intact muscularis for confident T1–T2 prediction
Illustration
The image shows a cT1–2 distal rectal tumor with a clearly visible, intact hypointense muscularis propria.
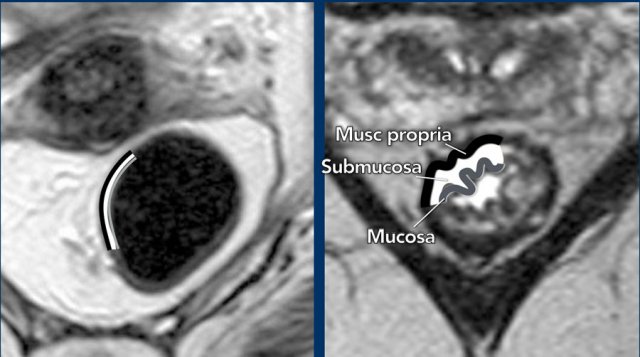
Differentiating T1 from T2 on MRI
Rectal Wall Anatomy:
- Inner layer: Mucosa (intermediate T2 signal)
- Middle layer: Submucosa (high T2 signal)
- Outer layer: Muscularis propria (low T2 signal)
MRI Appearance:
- Without edema: The rectal wall usually appears as two layers (muscularis propria distinguishable; mucosa and submucosa not reliably separable, which means that the distinction between T1 and T2 cannot be made (left image).
- Three-layer visualization is typically mainly possible with submucosal edema (right image).
T1 vs. T2 Differentiation:
- Some studies suggest high-resolution MRI may distinguish T1 (submucosal invasion) from T2 (muscularis invasion) and even assess submucosal depth (Sm1/2/3) .
- However, current guidelines (including updated ESGAR recommendations) still question MRI’s accuracy and reproducibility for this distinction.
- Practical approach: Tumors confined to the rectal wall are generally classified as T1–2 on MRI.
T3 Tumors
T3 tumors extend beyond the muscularis propria into the mesorectal fat.
MRI findings:
- Interruption of the hypointense muscularis propria
- Spicular or nodular tumor signal extension into the mesorectal fat
Image
This partly annular tumor (8–12 o’clock) invades the perirectal fat, with an anterior MRF margin <1 mm (arrow), consistent with cT3ab MRF+.
- Intact muscularis propria is visible from 12 to 8 o’clock
- Right-sided interruption of the muscularis, with nodular tumor extension into the perirectal fat (arrowheads)
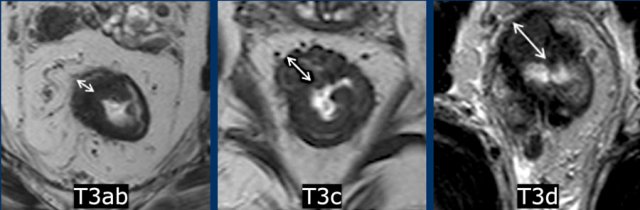
Subclassification
of T3 stage according to invasion depth
While the prognostic and therapeutic relevance of T3 subclassification remains debated, the ESGAR structured reporting template continues to use this classification:
Early T3:
- T3a: <1 mm extension beyond muscularis propria
- T3b: 1–5 mm extension
Advanced T3:
- T3c: 5–15 mm extension
- T3d: >15 mm extension
Key Consideration
Any T3 tumor with MRF involvement—regardless of subclassification—is always considered high-risk.
Pitfall: Desmoplastic Stranding vs. Tumor Invasion
Challenge
Distinguishing true perirectal tumor invasion (T3, Case A) from desmoplastic stranding (T1–2, Case B) can be impossible and is a common cause of overstaging.
Note
The distinction between T2 and early T3 (cT3ab) tumors is now considered less clinically relevant in current international guidelines.
Many guidelines group T2 and limited T3 (cT3ab) tumors together in the same favorable prognostic category for treatment stratification.
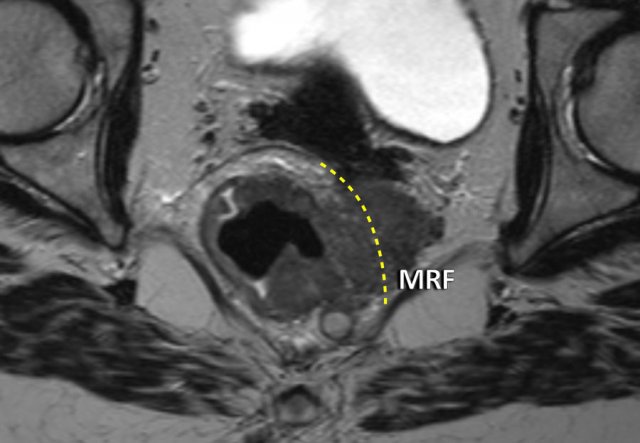
Mesorectal fascia (MRF) involvement
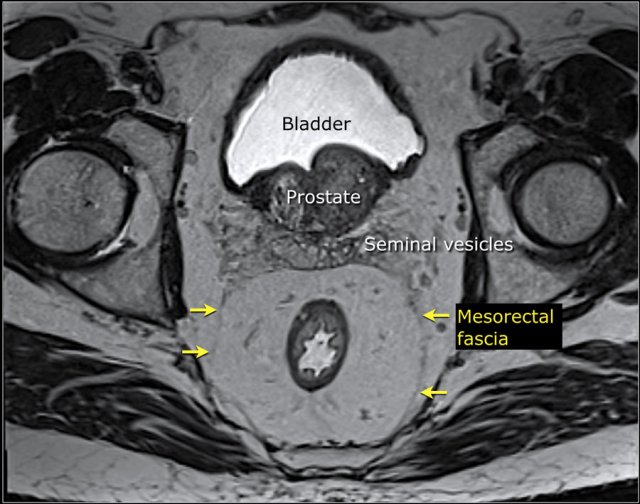
The MRF is a thin fibrous structure enclosing the mesorectal compartment and defines the anticipated resection plane during total mesorectal excision (TME).
On T2-weighted MRI, it appears as a thin hypointense line surrounding the mesorectum.
MRF Involvement Criteria:
- Direct tumor invasion of the MRF
- Tumor-MRF margin ≤1 mm
Clinical Implications
MRF involvement increases the risk of irradical resection and local recurrence with standard TME.
Neoadjuvant treatment is indicated to achieve tumor downsizing and MRF retraction, ensuring a tumor-free resection margin.
Reporting Requirement
Always specify the location of MRF involvement (e.g., “MRF+ at … o’clock” or “MRF+ at the left anterior side”).
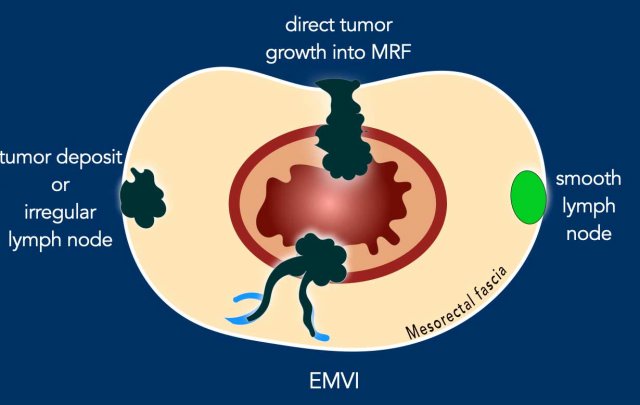
MRF Involvement by Tumor-Bearing Structures
MRF+ Criteria: Report MRF involvement if there is a ≤1 mm margin from:
- The primary tumor
- Extramural venous vascular invasion (EMVI)
- Tumor deposits or irregular lymph nodes (i.e. nodes with extracapsular extension)
Exception
- Smooth, enlarged lymph nodes with an intact capsule contacting the MRF have a very low risk of margin involvement on histopathology and do not significantly increase local recurrence risk.
- Do not classify these as MRF+ to avoid overtreatment.
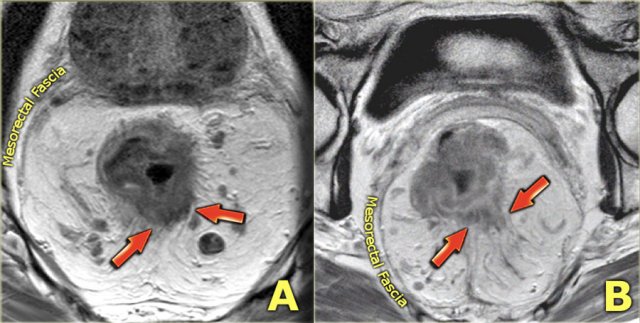
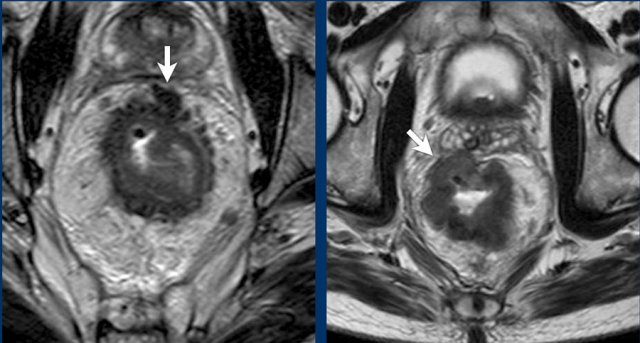 Two examples of T3 tumors with invasion of the mesorectal fascia. In the left case the distance between the tumor and the MRF is less than 1 mm at 12 o’clock. In the right case there is more extensive involvement of the MRF between 10 and 12 o’clock
Two examples of T3 tumors with invasion of the mesorectal fascia. In the left case the distance between the tumor and the MRF is less than 1 mm at 12 o’clock. In the right case there is more extensive involvement of the MRF between 10 and 12 o’clock
Circumferential Resection Margin (CRM) vs. Mesorectal Fascia (MRF)
Radiology Reporting
For T3+ tumors, always report the smallest tumor-MRF distance.
Some reports use the term "circumferential resection margin" (CRM) interchangeably with MRF, but this is not fully accurate.
Key Distinction:
- MRF: Thin fibrous boundary enclosing the mesorectum, visualized on MRI.
- CRM: The actual surgical margin created during TME, assessed by the pathologist.
- Ideally, CRM follows the MRF.
- If MRF is breached during surgery, CRM may be smaller.
- If extra fat/organs are included, CRM may be wider.
Note: Use "MRF" in radiology reports to avoid confusion with the surgical/pathological CRM.
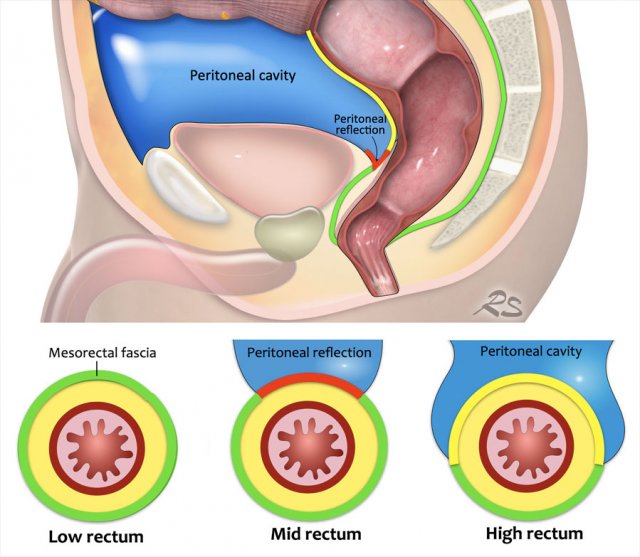
Mesorectal fascia vs. Peritoneum
Anatomical Coverage:
- Low rectum: Fully enclosed by the mesorectal fascia (MRF, green line).
- Middle rectum:
- Posterior/lateral: Covered by MRF
- Anterior: Covered by peritoneum (red line, peritoneal reflection)
- High rectum: Peritoneum extends anteriorly and laterally (yellow line); MRF covers only the dorsal mesorectum.
Clinical Significance:
- MRF invasion: Classified as T3 MRF+
- Peritoneum invasion: Risks peritoneal tumor spread; staged as T4a
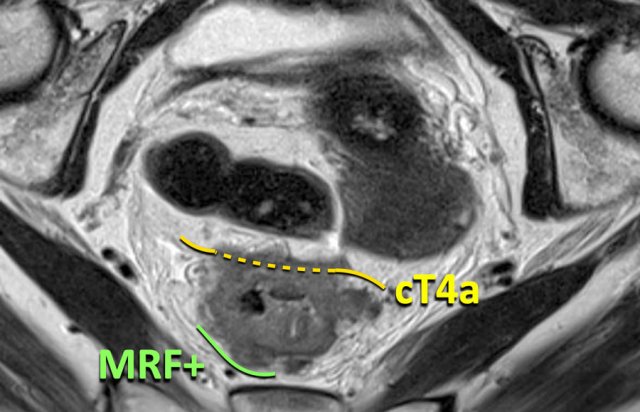
T4a Tumor
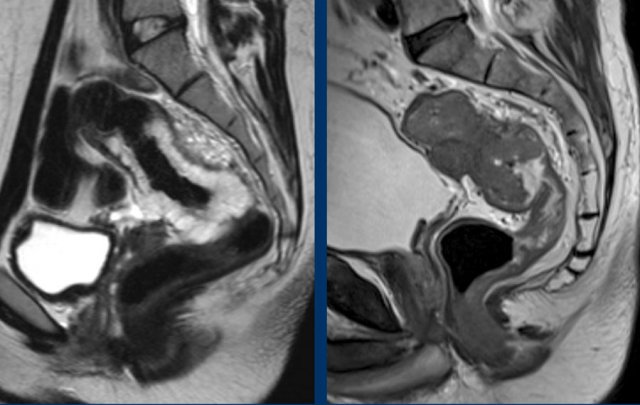
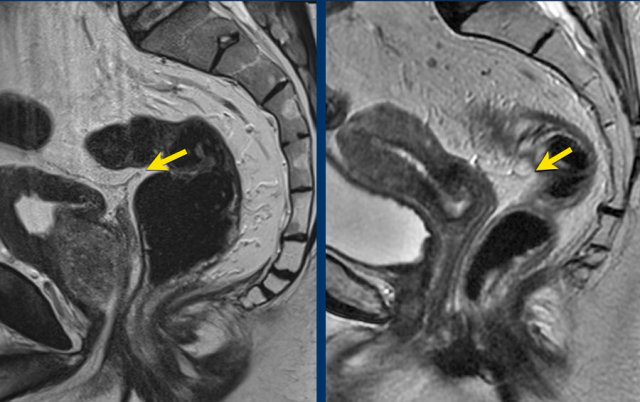
Anatomical Landmark:
- The anterior peritoneal reflection demarcates the transition between the non-peritonealized and peritonealized rectum.
- Below this reflection, the rectum is entirely extraperitoneal.
MRI Identification:
- On sagittal T2-weighted images, the peritoneal reflection appears as a hypointense, V-shaped thin line (arrow).
- Location:
- Males: Just above the seminal vesicles
- Females: At the level of the cul-de-sac (Douglas pouch)
Staging Implication:
- Anterior invasion of the peritoneal reflection or peritoneum defines T4a disease.
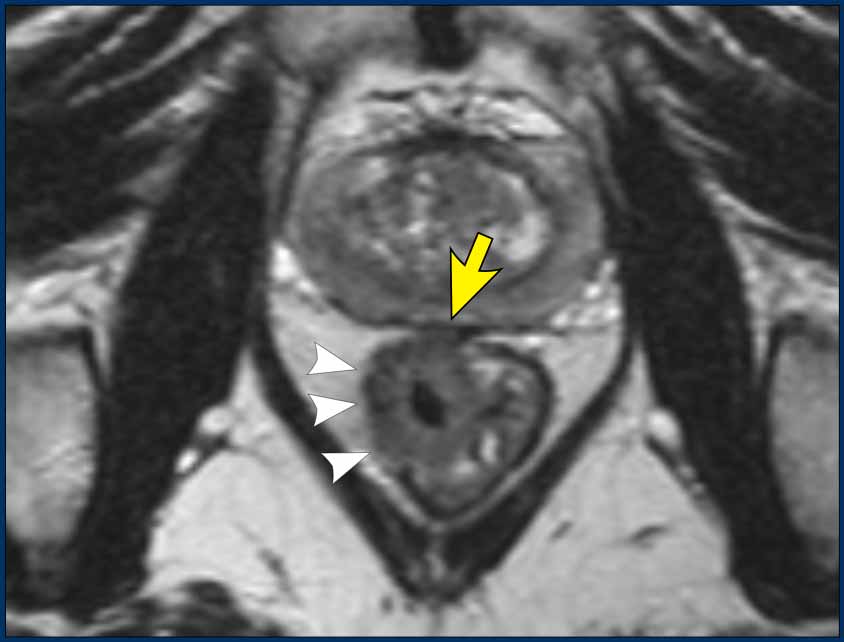
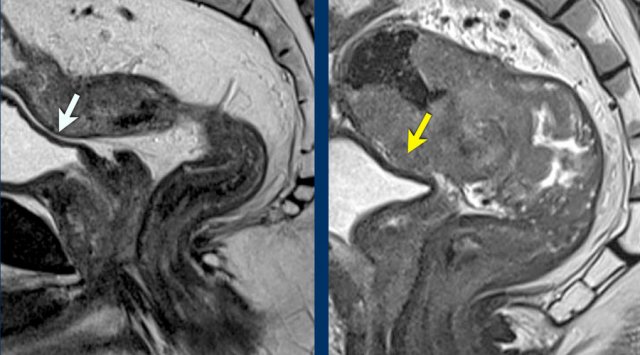
Pitfall: Avoiding Overstaging of Peritoneal Invasion in High Rectal Tumors
Key Point:
In proximal rectal tumors, the rectum and peritoneum are often in close anatomical proximity.
This alone does not indicate T4a disease.
Diagnostic Criteria for cT4a:
- Actual tumor signal extension into or beyond the peritoneum/peritoneal reflection
- Peritoneal thickening caused by the tumor
Image Examples:
- Left: Tumor in close proximity to the peritoneum and bladder (white arrow), but no extension beyond the muscularis propria or into the peritoneum—not T4a.
- Right: Tumor signal extends into the peritoneum (yellow arrow)—confirms T4a invasion.
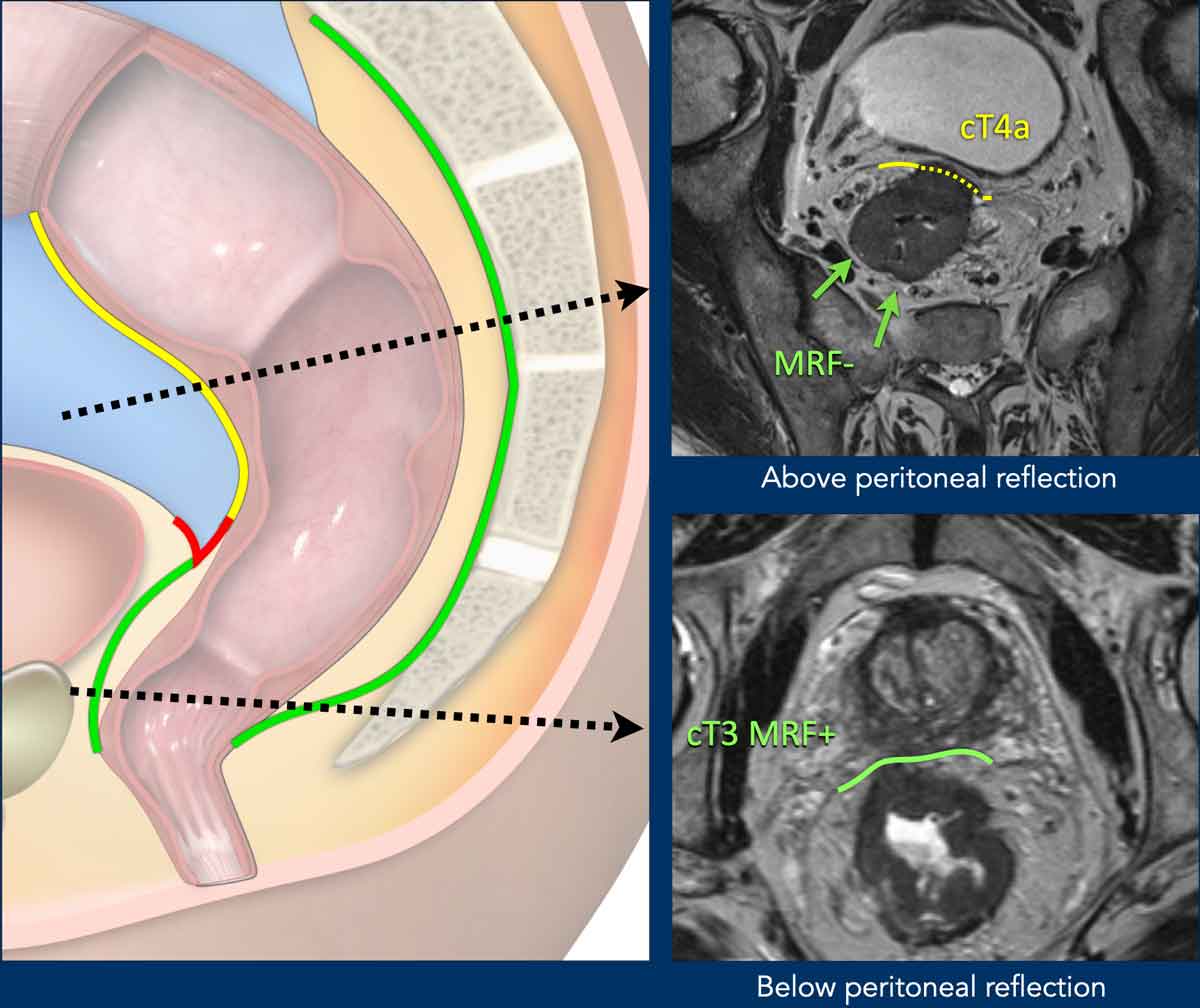
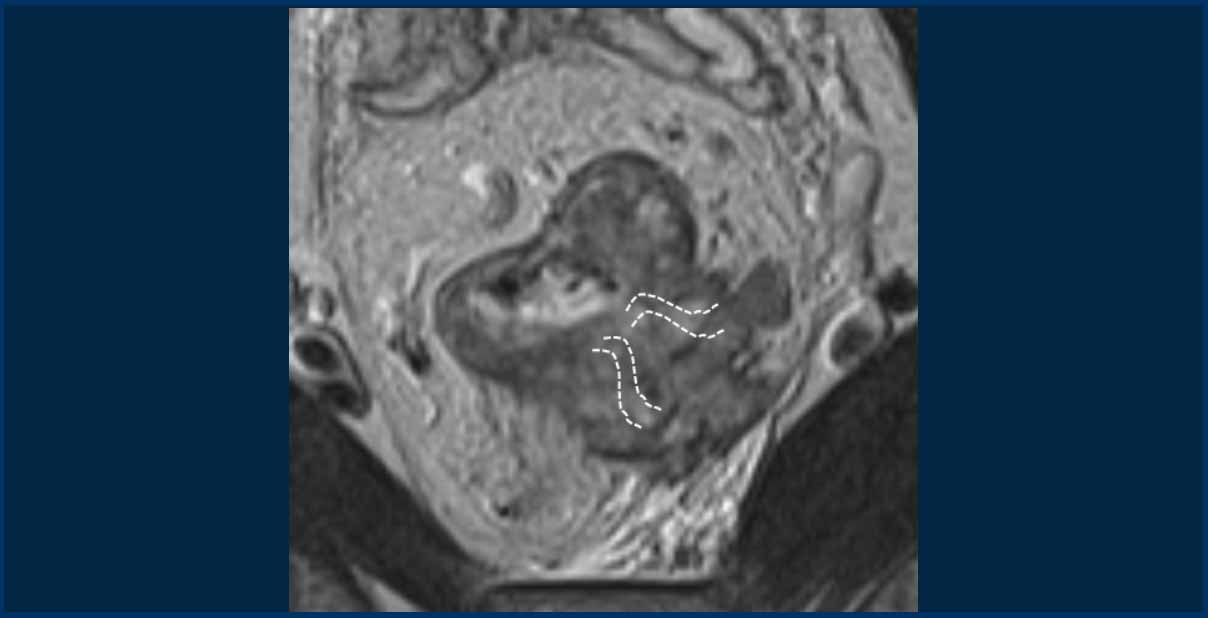
Pitfall: Distinguishing MRF vs. Peritoneal Invasion in Anterior Tumors
Key Principle:
- Anteriorly, MRF invasion can only occur in tumors below the peritoneal reflection, reported as T3 MRF+
Anterior tumors above the peritoneal reflection that invade the peritoneum should be reported as T4a MRF - Posteriorly, the MRF extends above the level of the peritoneal reflection. Simultaneous involvement of the MRF (posteriorly) and peritoneum (anteriorly) can occur, which should be reported as T4a MRF+
Image Examples
cT4a MRF– (Upper Image):
- Upper rectal tumor, above the peritoneal reflection
- Peritoneal involvement (dashed yellow line)
- No MRF involvement
cT3 MRF+ (Lower Image):
- Distal rectal tumor, below the peritoneal reflection
- Anterior MRF involvement (green line)
- No peritoneal involvement
cT4a MRF+ Tumor
This proximal rectal tumor demonstrates combined involvement:
- Anterior: Peritoneal invasion (T4a)
- Posterior: Mesorectal fascia (MRF) invasion (MRF+)
T4b tumors
T4b tumors invade organs, anatomical structures, or compartments outside the mesorectum (red area), excluding:
- Mesorectal fascia (classified as T3 MRF+)
- Peritoneum/peritoneal reflection (classified as T4a)
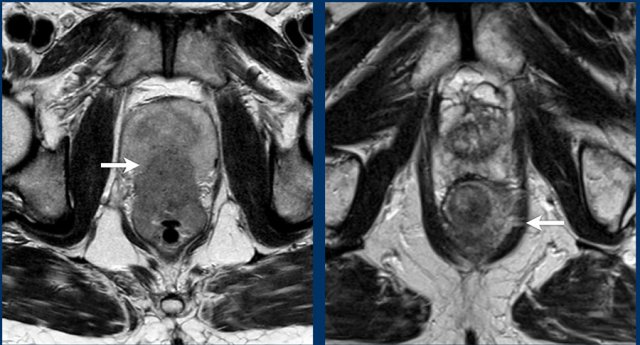
Examples of cT4b Disease
- Left: Tumor invading the prostate
- Right: Tumor invading the left puborectalis muscle
Note
Invasion of striated muscles (e.g., external anal sphincter, puborectalis, levator ani) is classified as cT4b disease.
cT4b Tumor Example
This image demonstrates a tumor extending beyond the mesorectal compartment and MRF (likely located at the level of dashed line) into the left obturator space fat.
Conclusion:
cT4b tumor.
T4b Invasion: Diagnostic Criteria and Management Implications
T4b invasion is confirmed when tumor signal extends into an adjacent organ or structure, characterized by:
- Tumor signal extending into adjacent organ or structure
- Loss of the separating fat plane
- Disruption of the normal organ contour
Indeterminate Findings:
If there is loss of the fat plane but no clear organ contour disruption or definitive tumor signal extension, report as:
- Focal/broad-based contact
- Potential (but not definitive) invasion
Surgical Implications:
- Advanced T4b disease often requires beyond-TME surgery (e.g., pelvic exenteration), with en bloc resection of the mesorectum and adjacent organs.
Note that ESGAR/ESUR/SAR/PelvEx have published separate guidelines for MRI reporting of advanced (and recurrent) tumors before pelvic exenteration, using an anatomy-based template [ref].
These include practical recommendations on how to assess specific structure involvement::
- Vessels: Luminal narrowing/occlusion, thrombus, or collaterals
- Ureters: Upstream dilatation (hydroureter/hydronephrosis)
- Nerves: Thickening compared to the contralateral side
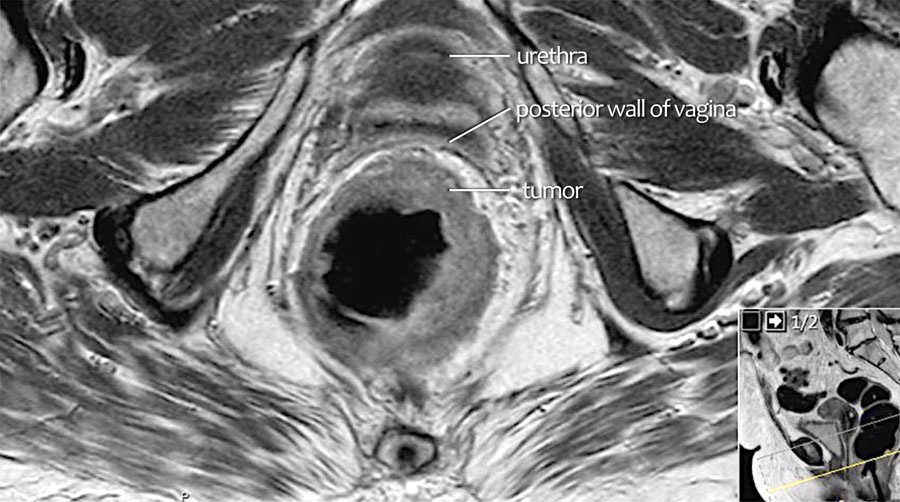
Example
Scroll through the images.
On axial MRI, intermediate tumor signal extends into the posterior vaginal wall (arrows) indicating T4b invasion.
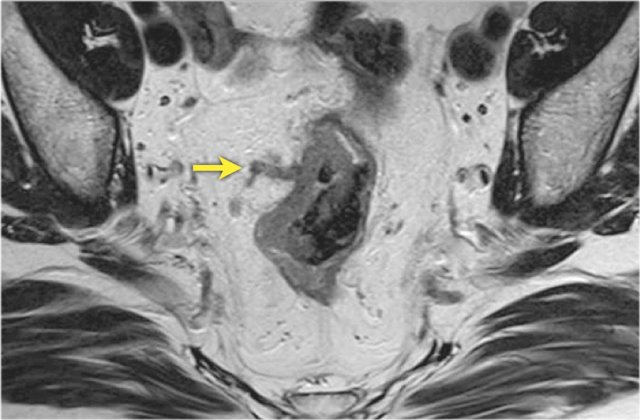
Extramural vascular invasion (EMVI)
EMVI is a risk factor for recurrent disease, metastases and impaired overall survival.
EMVI is suspected if we see tumor-signal extending into an adjacent vessel, when the vessel is expanded by tumor, or if the tumor disrupts the vessel borders (illustration).
EMVI can be graded as follows [ref]:
- 0 = no nodular extension beyond the muscularis propria and no vessels adjacent to areas of tumor extension
- 1 = Minimal nodular extension or stranding, not in the vicinity of vascular structures
- 2 = stranding in the vicinity of extramural vessels, with vessel of normal caliber and no tumor signal extending into the vessels
- 3 = tumor signal extending into vessels, with normal or slightly expanded vessel contour
- 4 = tumor signal extending into vessels with clear disruption of vessel contour and/or nodular expansion of the vessels
Grades 3 and 4 should be reported as EMVI+ disease.
Grades 0-2 as EMVI-.
Example
This image demonstrates EMVI+ disease, showing tumor signal extending into an adjacent vessel, resulting in vessel expansion and contour disruption.
Pitfall: Assessing EMVI in tumors with massive extramural growth
In large T3cd-4 tumors with extensive extramural extension into the perirectal fat, a practical approach is to classify these as EMVI+.
In the example shown, the tumor extends into the perirectal fat from 3 to 5 o’clock, obliterating the mesorectal fat in this region.
While individual vessel involvement may not be discernible, it is reasonable to assume that vessels in this area are completely invaded by the tumor.
The dashed lines indicate the potential location of vessels.
Pitfall: overstaging of EMVI
Normal perirectal vessels of varying diameters often radiate outward from the muscularis propria into the mesorectal fat.
Do not misinterpret these as EMVI unless there is clear tumor invasion—defined as tumor signal extending into, expanding, or disrupting the vessel contour.
Key Assessment Tip
Evaluate the tumor’s circumferential location within the rectal wall.
Vessels not originating from the tumor attachment site should never be classified as EMVI, regardless of prominence.
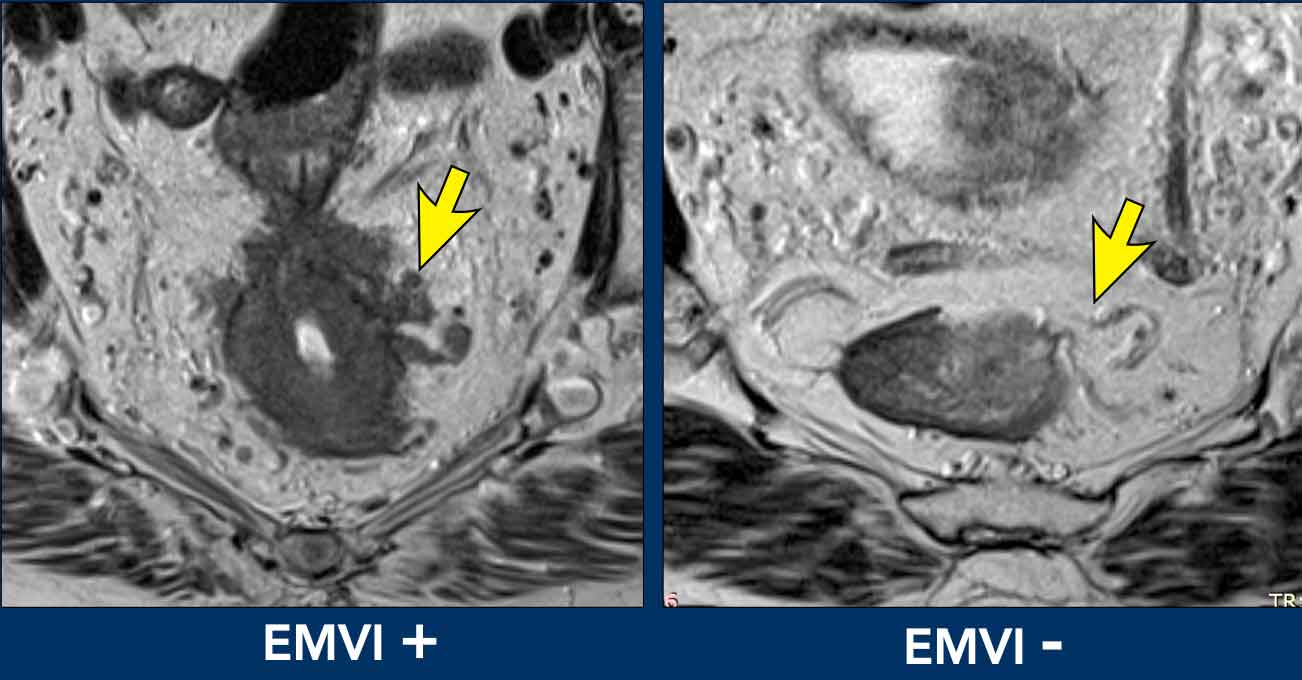
Image comparison
- Left (EMVI+): Tumor signal clearly extends into adjacent vessels, disrupting their contours.
- Right (EMVI-): Vessels maintain smooth contours with no tumor signal present.
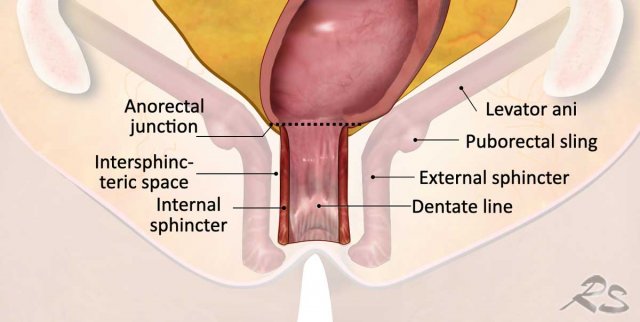
How to report anal sphincter and pelvic floor involvement
The anal sphincter complex consists of three layers:
- Internal sphincter: Continuation of the rectal smooth muscle
- Intersphincteric space: Located between the internal and external sphincters
- External sphincter: Voluntary striated skeletal muscle, continuous cranially with the puborectalis and levator animuscles
The puborectalis and levator ani, together with the iliococcygeus and pubococcygeus, form the pelvic floor.
MRI Reporting for Low Rectal Cancers
For surgical planning, the MRI report must clearly describe:
- Tumor relationship to the anal sphincter and pelvic floor
- Layers involved (internal/external sphincter, pelvic floor muscles)
- Location (left/right)
- Extent of invasion: upper, middle, or lower third of the anal canal
Explicitly indicate anal involvement, e.g., by adding the suffix ‘anal+’.
Anal sphincter involvement and cT-category
The TNM staging system does not specify how invasion of the anal sphincter or pelvic floor layers should be classified.
Current guidelines recommend:
- Base cT-category on the extent of tumor invasion within the rectum.
- Exclude involvement of the internal sphincter and intersphincteric plane from cT categorization.
- Classify invasion of the external sphincter, puborectalis, or levator ani muscles (i.e., skeletal muscle invasion) as cT4b disease.
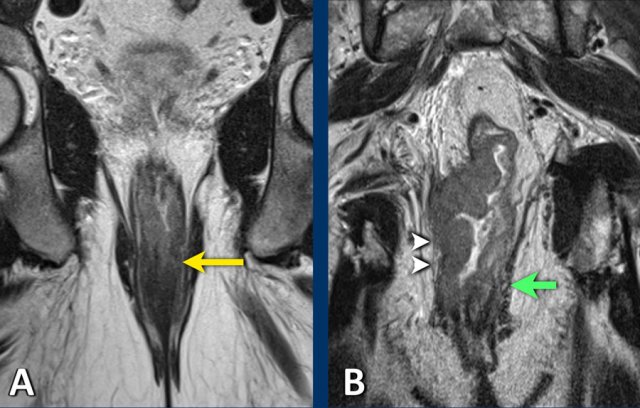
Examples
A. cT1-2 low rectal tumor invading the internal sphincter (left, yellow arrow).
Note: Internal sphincter invasion does not upstage cT-category but must be explicitly reported to guide surgical planning.
B. Distal tumor invading both internal and external sphincters (right side, arrowheads), with involvement of the puborectalis and levator ani muscles. The invasion of the external sphincter, puborectalis, or levator ani constitutes skeletal muscle invasion and should be classified as cT4b disease.
There is a normal appearance of the external sphincter and pelvic floor muscles on the left side in B (green arrow).
N-category
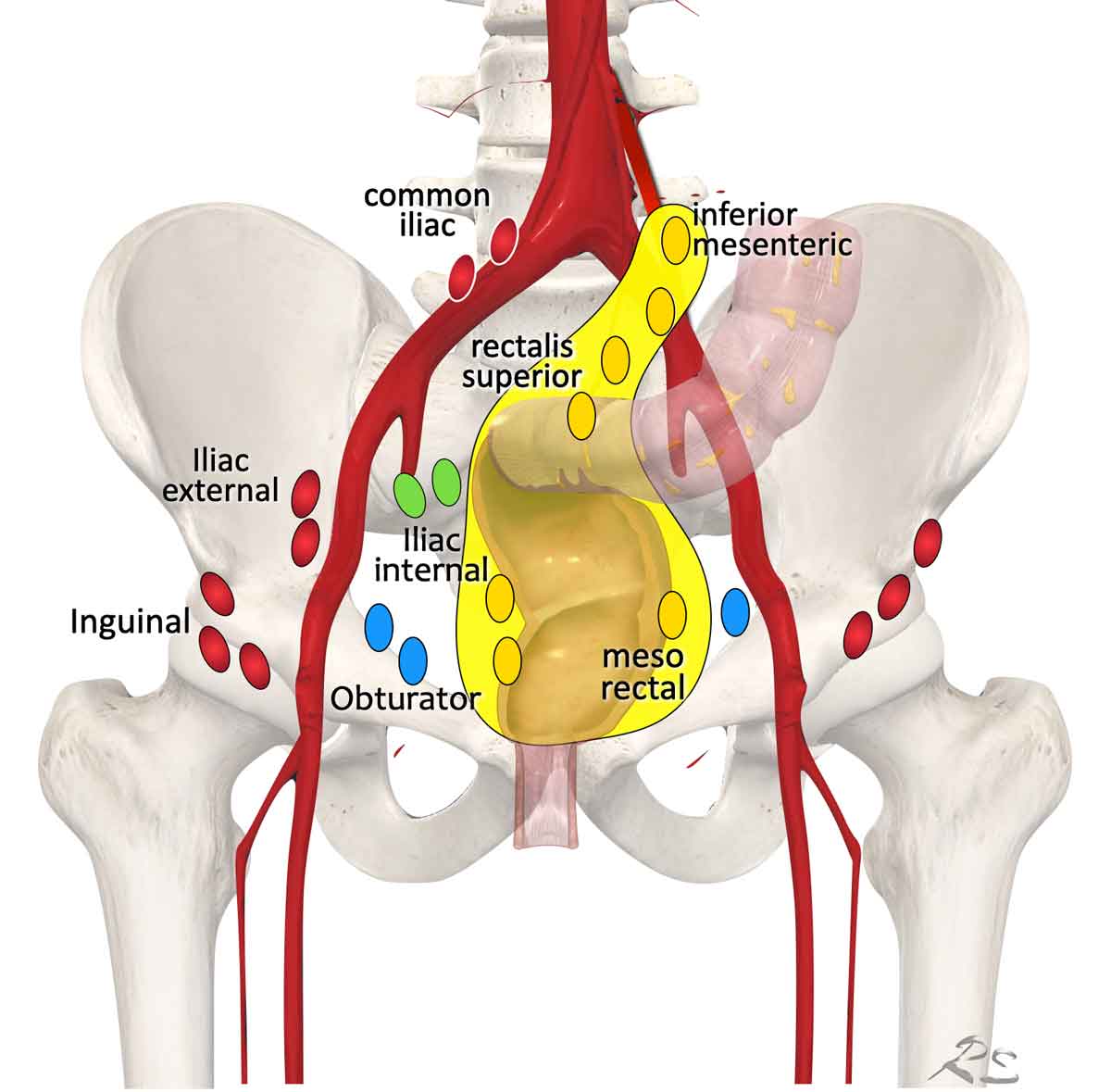 Regional lymph node drainage. The lymph nodes in red are all non, regional (M-stage) nodes. In TME only the mesorectal nodes and in high rectal tumors also the rectalis superior and inferior mesenteric nodes are excised.
Regional lymph node drainage. The lymph nodes in red are all non, regional (M-stage) nodes. In TME only the mesorectal nodes and in high rectal tumors also the rectalis superior and inferior mesenteric nodes are excised.
Lymph Node Map
Terminology for lymph node stations in rectal cancer can be confusing, with inconsistent use of terms like ‘extramesorectal nodes’ and ‘lateral nodes’ to describe both regional and non-regional lymph node stations.
For accurate treatment planning, distinguish between:
Regional Lymph Nodes (cN-category)
- Mesorectal nodes (yellow): Include ‘high mesorectal lymph nodes’ (up to the origin of the inferior mesenteric artery), located along the presacral inferior mesenteric and superior rectal vessels.
- Obturator nodes (blue)
- Internal iliac nodes (green)
Non-Regional Lymph Nodes (cM-category)
Lymph nodes outside the cN-category are considered distant metastases (cM):
- External iliac nodes
- Common iliac nodes
- Inguinal nodes
Radiotherapy/Surgical Planning
Standard TME removes only mesorectal nodes.
The upper margin of TME is defined by tumor level and the most proximal suspicious lymph node (i.e., whether nodes along the inferior mesenteric artery are included).
Explicitly report involvement.
Obturator and internal iliac nodes, though cN-category, are not routinely excised—these may require targeted radiotherapy or lateral lymph node dissection to reduce lateral recurrence risk.
Exception: AJCC TNM (8th ed.) classifies inguinal nodes as regional for low rectal tumors extending into the distal anal canal (below the dentate line), as these follow the anal canal’s regional lymphatic drainage.
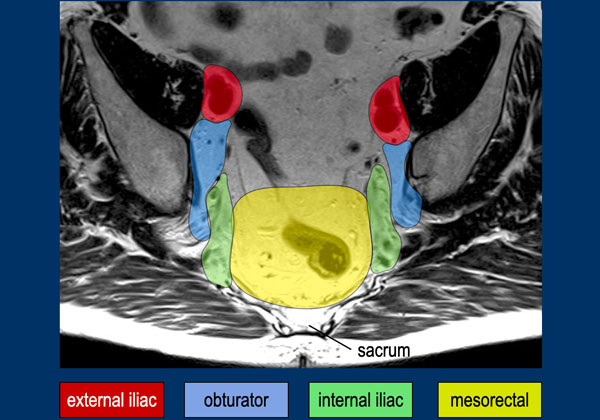
This MR image demonstrates the mesorectal, internal iliac, obturator, and external iliac lymph node compartments, as outlined by the Lateral Node Study Consortium [ref].
Key Points:
- External iliac nodes are non-regional; if positive, they are classified as metastatic disease (cM).
- The obturator and internal iliac compartments are separated by the lateral border of the main trunk of the internal iliac vessels.
- The posterior border of the external iliac compartment is defined by the posterior border of the external iliac vessels.
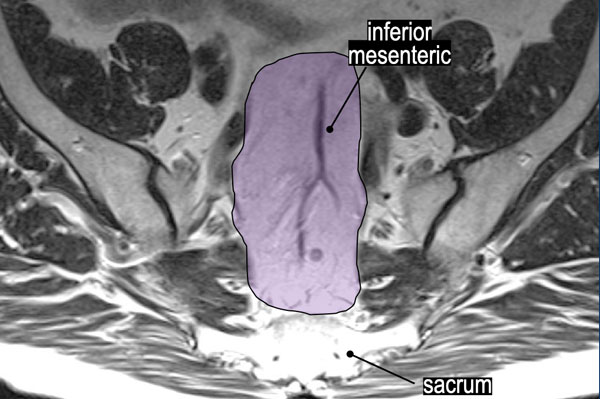
Superior Rectal and Inferior Mesenteric Lymph Nodes
The purple area highlights the superior rectal and inferior mesenteric lymph nodes ("high mesorectal nodes"), which are regional N-stage nodes.
Reporting Requirements:
- Specify the level of the most proximal suspicious node in this region (e.g."Most proximal lymph node located along the presacral blood vessels at the S2 level.").
- Note any suspicious nodes along the root of the inferior mesenteric vessels.
Clinical Impact: This information directly influences radiotherapy field planning and surgical resection strategy.
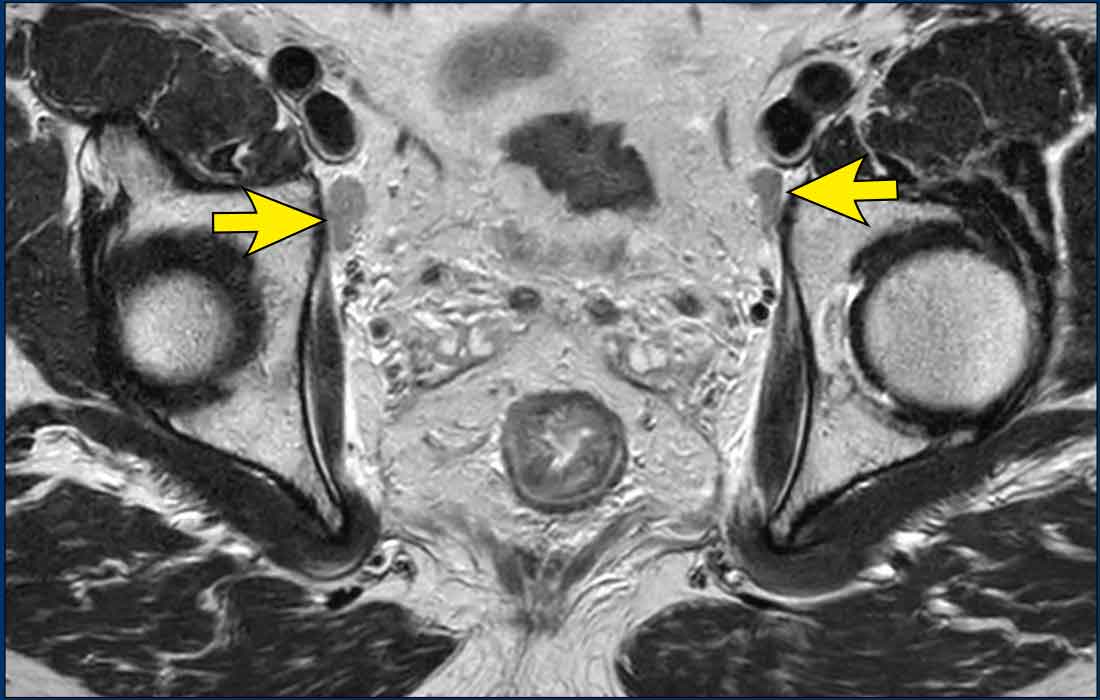
Pitfall – benign nodes dorsal from the external iliac veins
Nodal metastases along the external iliac vessels are rare in rectal cancer and, if present, indicate metastatic disease (cM).
However prominent benign lymph nodes located immediately dorsal to the external iliac veins are a common finding and should not be mistaken for metastases.
These nodes typically have an oval or elongated shape and frequently occur bilaterally (figure).
Their morphology is characteristic of benign reactive lymph nodes, and in most cases, they can be safely omitted from the report.
The 7-mm short-axis cutoff used for assessing obturator and internal iliac nodes is not applicable to external iliac nodes, as reactive nodes in this region frequently exceed 7 mm in short-axis diameter.
Criteria for Suspicious Lymph Nodes
MRI’s limited accuracy for N-staging has shifted focus toward more reliable risk factors (e.g., MRF invasion, EMVI). The ESGAR guidelines now advocate a risk-adaptive approach over detailed node-by-node assessment.
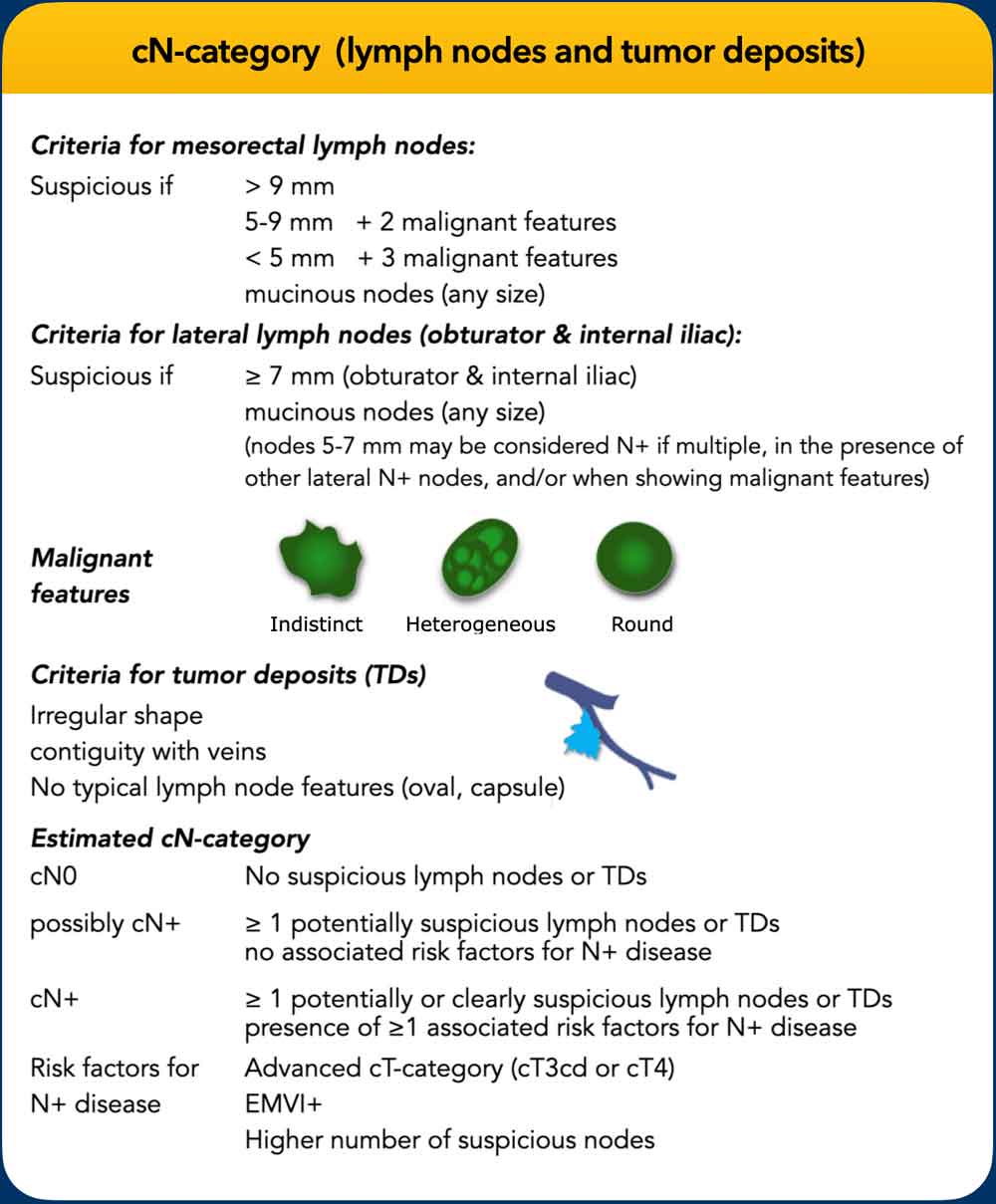
cN-Category Criteria
- Mesorectal nodes:
- Suspicious if: ≥9 mm or mucinous signal.
- Smaller nodes: Require additional malignant features for cN+ classification.
- Dutch guidelines: Nodes <5 mm (unless mucinous) are considered benign.
- Lateral nodes (internal iliac/obturator):
- Suspicious if: ≥7 mm; malignant morphology supports cN+ in intermediate-sized nodes.
- Tumor deposits:
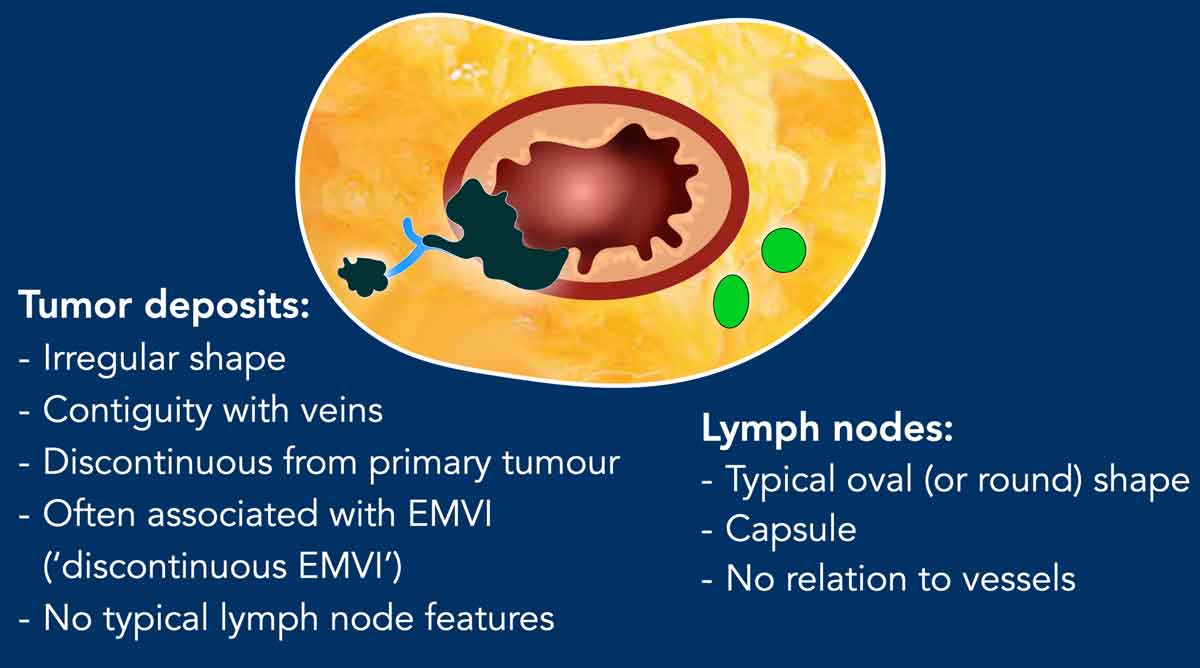
- Distinguishing features: Irregular shape, vein contiguity, lack of typical lymph node morphology (e.g., oval shape, capsule).
- Distinguishing features: Irregular shape, vein contiguity, lack of typical lymph node morphology (e.g., oval shape, capsule).
Traditional vs. Current cN Staging
- Traditionally: N0 (0 nodes), N1 (1–3 nodes), N2 (≥4 nodes), with further subclassifications (N1abc, N2ab).
- Current practice: Simplified to cN0/N+ due to MRI’s limited accuracy.
- ESGAR recommendation: Include a confidence level, considering additional risk factors for nodal involvement.
Practical cN Classification
- cN0: No suspicious lymph nodes.
- Possibly cN+: Borderline or ambiguous nodes (especially if multiple).
Downstage to cN0 if no other high-risk features
Upgrade to cN+ if additional risk factors are present. - cN+: Clearly pathologic nodes or suspicious nodes with additional risk factors.
Additional Risk Factors: Advanced T-category, EMVI, multiple suspicious nodes.
Lateral lymph nodes (internal iliac/obturator)
Internal iliac and obturator lateral lymph nodes are primarily assessed by size, with ≥7 mm (short axis) as the main criterion for cN+.
Morphologic features—indistinct borders, heterogeneous signal, round shape, and loss of fatty hilum—can support malignancy in intermediate-sized (5–7 mm) nodes, especially if multiple or in the presence of other clearly pathologic lateral nodes.
Reporting Requirement
Any lateral N+ nodes (though included in the overall cN-category) must be highlighted separately in the MRI report, with location and size specified, as these require targeted treatment to prevent lateral recurrence.
Images
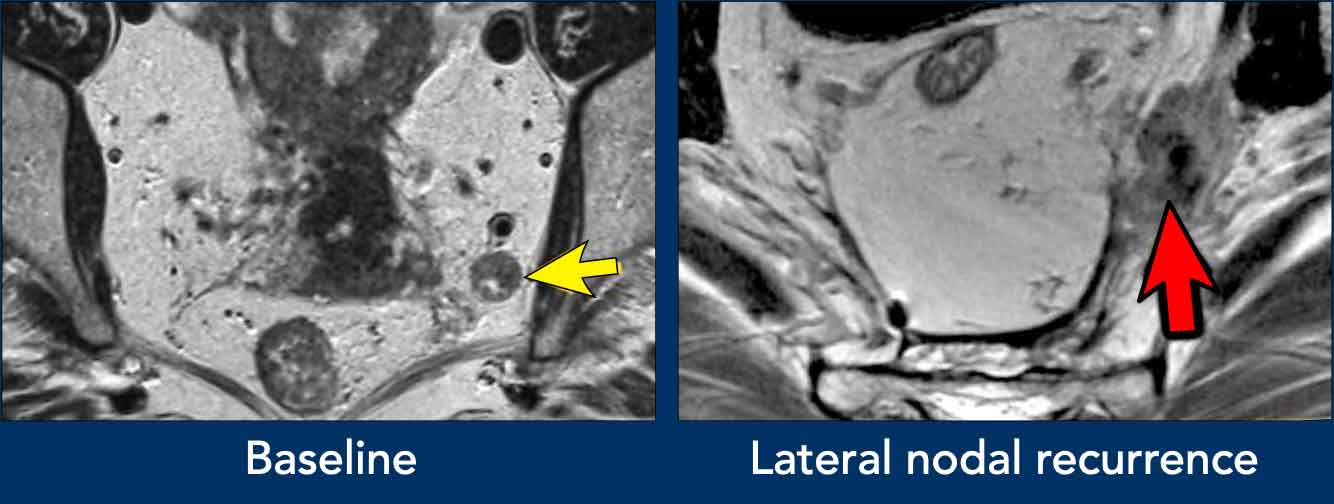
- Left image: Pathologic 9 mm lymph node in the left obturator space (arrow), identified at baseline staging. Requires irradiation and/or separate resection to avoid lateral recurrence.
- Right image: Patient treated with standard TME (without lateral nodal resection) who developed an irregular mass in the left obturator space, consistent with lateral nodal recurrence.
Tumor Deposits
Tumor deposits are recognized as a high-risk feature with poorer prognosis than pathologic lymph nodes.
While MRI’s reliability in distinguishing tumor deposits from lymph nodes remains debated, ESGAR guidelines suggest irregular shape and venous contiguity as key imaging criteria.
In contrast, lymph nodes are typically oval, encapsulated, and not vessel-associated.
Reporting: Although tumor deposits are classified under the cN-category, their suspected presence must be explicitly noted in the MRI report.
N1c Category (TNM 8th Ed.)
- Definition: Tumor deposits without pathologic lymph nodes.
- Challenge: Differentiating small tumor deposits from lymph nodes remains difficult; large-scale validation of imaging criteria is lacking.
- ESGAR Recommendation: Due to limited reliability on MRI, avoid subclassifying as N1c.
Instead, combine tumor deposits and lymph nodes under a simplified cN-category (cN0, possibly cN+, or cN+).
Tumor deposits and EMVI
Tumor deposits are strongly associated with EMVI, often considered a continuation of vascular tumor spread—beginning with EMVI and progressing to tumor deposits in advanced disease [ref].
MRI data show that tumor deposits co-occur with EMVI in up to 80% of cases [ref].
Image Example
- White arrows: EMVI—tumor signal extending into adjacent vessels, with vessel expansion and contour disruption.
- Black circles: Tumor deposits seeded along the vessels.
M-stage
The M-stage in rectal cancer is based on the presence of suspicious non-regional lymph node metastases and other distant metastases.
Note that non-regional lymph nodes are together considered as one “organ”.
Restaging after neoadjuvant treatment
Checklist
In the era of organ preservation (watch-and-wait), the primary goal of restaging is to stratify patients by response to neoadjuvant treatment, guiding further management.
The ESGAR guidelines [ref] classify response as:
- Major residual tumor (poor response) → radical resection (TME) indicated
- Minor residual tumor (good response) → both TME and organ-preserving options (local excision) may be considered
- (Near-)complete response → organ preservation (watch-and-wait) may be appropriate. Endoscopy must be used alongside MRI to confirm response and guide treatment.
Note
- Detailed assessment of yT-category, yEMVI, yMRF, and sphincter invasion is only relevant if residual tumor is suspected.
- In (near-)complete responders and good responders with predominant fibrosis, accuracy of post-treatment MRI is poor due to fibrosis replacing the tumor bed, and hence of limited clinical value..
Key Reporting Points for Suspected Residual Tumor
Radiologists should highlight features that impact surgical strategy and may require beyond-TME surgery:
- ycT4b invasion
- Persistent sphincter or MRF involvement
- Suspicious lateral lymph nodes
Response assessment
After chemoradiotherapy, rectal tumors typically shrink and undergo fibrotic transformation, visible as marked T2 hypointensity in the tumor bed.
Differentiating fibrosis from viable residual tumor remains challenging.
Heterogeneous T2 signal or persistent mass effect within fibrosis raises suspicion for residual disease.
DWI significantly improves accuracy by detecting areas of restricted diffusion within fibrosis.
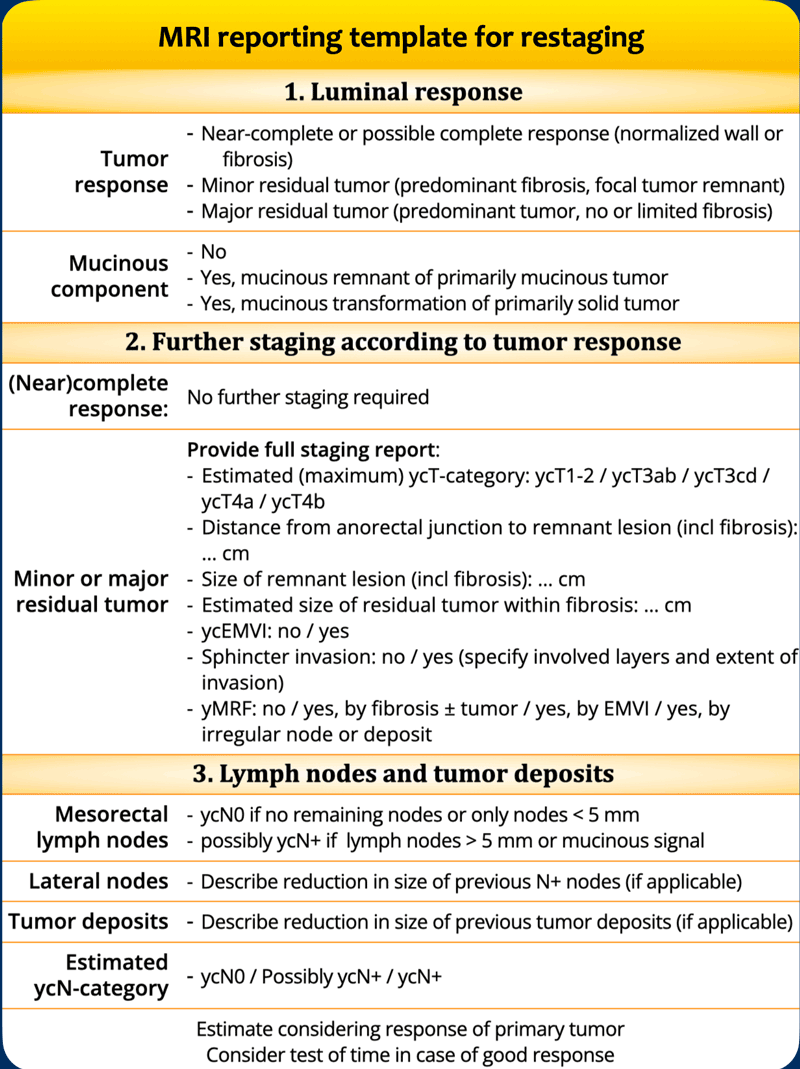
Restaging MRI Report Structure
Begin with a general description of:
- Degree of response (T2-weighted MRI morphology)
- Presence/absence of restricted diffusion on DWI
Response Classification
- (Near-)complete response – Potential candidates for watch-and-wait (if endoscopy confirms MRI findings)
- Minor residual tumor – Requires further treatment of the remnant (local excision or TME, stage-dependent)
- Major residual tumor – Generally necessitates radical resection (TME)
MR Tumor Regression Grade (mrTRG)
- Imaging adaptation of histopathological TRG, using a 5-point scale to grade fibrotic transformation on T2-weighted MRI (see table).
- Separate mrTRG reporting is not mandatory (per ESGAR guidelines), but mrTRG features (signal heterogeneity, tumor signal within fibrosis) complement DWI findings to refine response categorization.
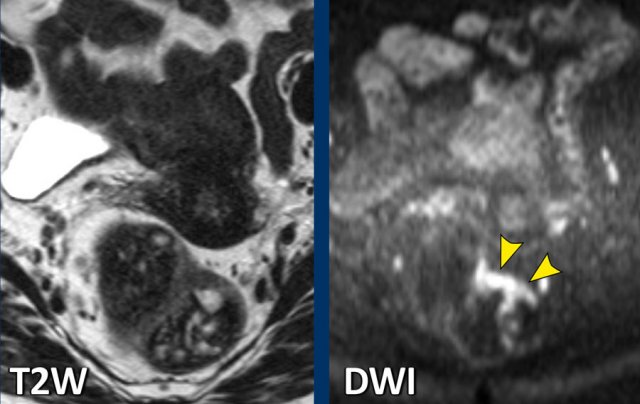
Role of DWI in detecting residual tumor
Diffusion-weighted imaging (DWI) highlights high-cellular-density tissue where extracellular water movement is restricted.
Its primary value lies in detecting residual tumor within focal (partially annular) fibrotic wall thickeningat the treated tumor bed.
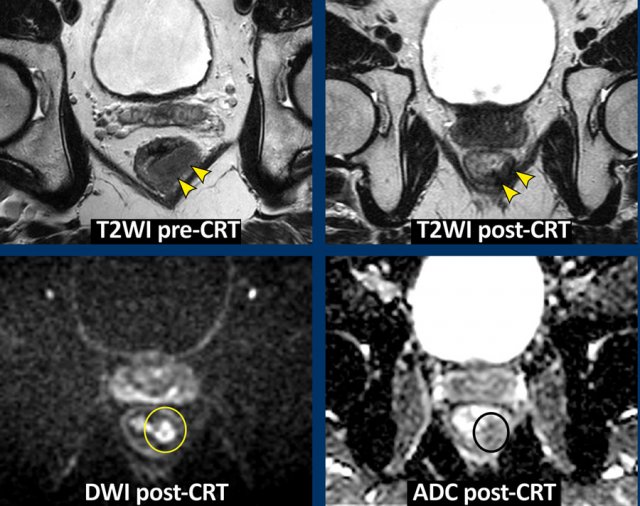
In cases of residual tumor, high signal is typically seen at the inner margin of the fibrosis on high b-value DWI, with correspondingly low signal on the ADC map.
Image Example
- Primary staging and restaging T2-weighted images show a predominantly fibrotic tumor bed with minor signal heterogeneity (mrTRG 3) post-chemoradiotherapy.
- Restaging DWI reveals a focal high-signal area at the inner margin of the fibrosis, with low ADC signal, indicating restricted diffusion.
- Histopathology confirmed this as a small tumor remnant (ypT2)
DWI pitfalls
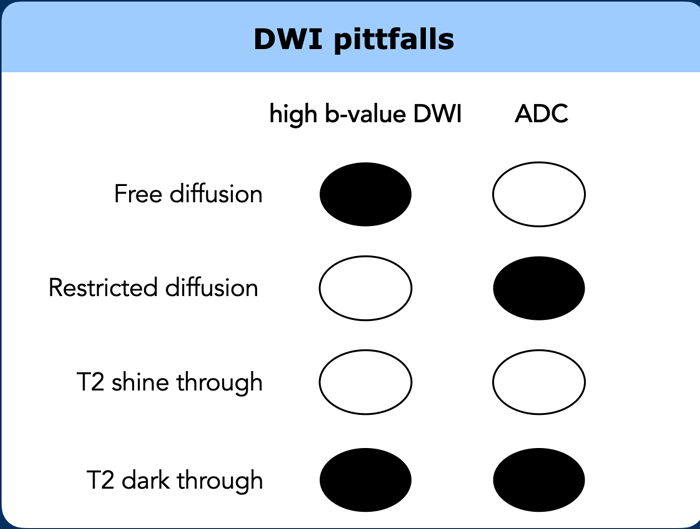
T2 Shine-Through
- Diffusion-weighted images (DWI) are inherently T2-weighted.
- T2 shine-through refers to high DWI signal caused by long T2-relaxation time (e.g., fluid), not restricted diffusion.
- In rectal DWI, small amounts of luminal fluid may mimic tumor signal in the adjacent rectal wall.
- Differentiation: n the ADC map (and T2W images), luminal fluid shows high signal, unlike tumor.
T2 Dark-Through (T2 Blackout)
- T2 dark-through appears as markedly low signal on the ADC map in areas of dense fibrosis without vital tumor.
- Occurs in tissues with very short T2-relaxation time (e.g., collagen-rich fibrosis, calcifications, cortical bone).
- Results in hypointense (black) signal on ADC, DWI, T2-, and T1-weighted sequences.
- Key distinction: T2 dark-through signal is distinctly black on ADC, whereas tumor appears grey.
- Not suspicious for tumor and should not be confused with restricted diffusion.
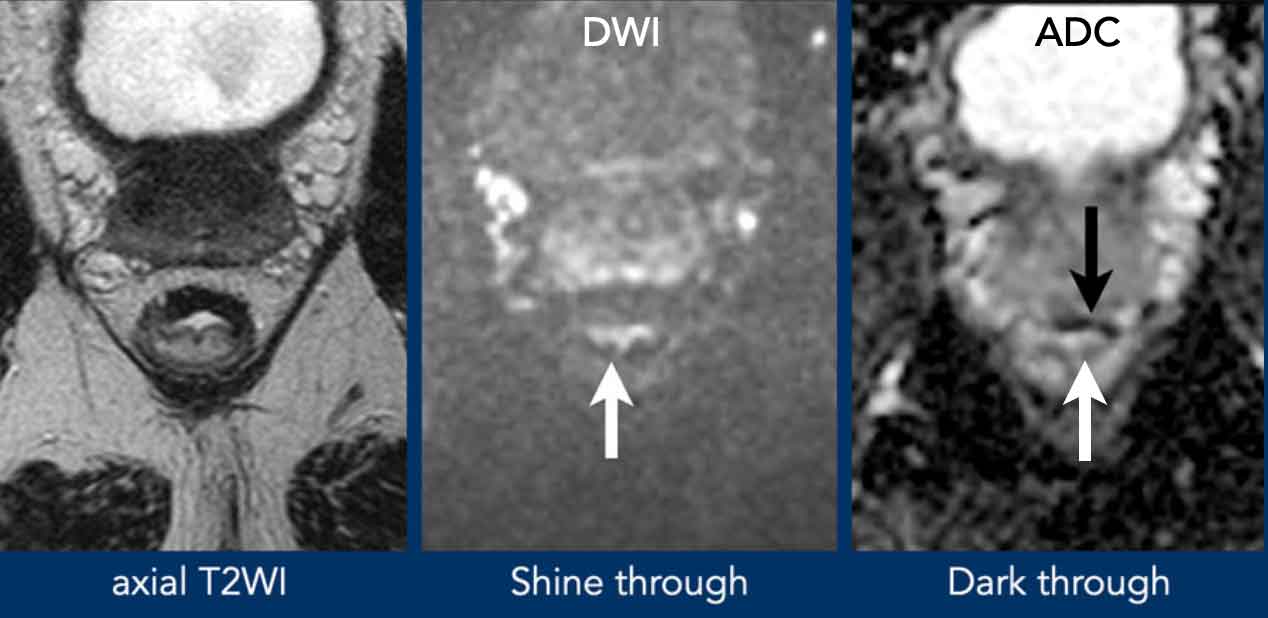
T2 Shine-Through
The white
arrow indicates high
DWI signal from fluid in the rectal lumen, confirmed by high signal on the
ADC map.
T2 Dark-Through
The black arrow shows distinctly low signal in the fibrotically changed anterior rectal wall on the ADC map.
No corresponding high DWI signal in the wall, confirming absence of true diffusion restriction.
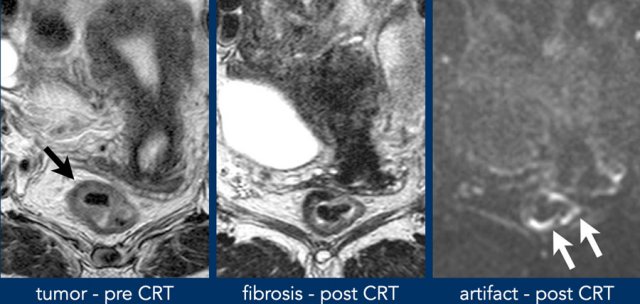
Susceptibility artefacts
Abdominal DWI scans are typically acquired using echo planar imaging (EPI), enabling rapid acquisition with minimal motion artifacts.
However, EPI-DWI is highly sensitive to susceptibility artifacts, particularly at higher field strengths.
These artifacts cause distortions or artificial signal pile-up at the rectal wall–gas interface, potentially mimicking tumor signal.
While large artifacts are easily recognized, subtle artifacts may be misinterpreted as pathology.
Current guidelines recommend a preparatory micro-enema for restaging (optional for primary staging) to minimize luminal gas and reduce susceptibility artifacts.
Images
- Pre CRT: CRT: tumor in the right anterolateral rectal wall (black arrow).
- Post-CRT fibrosis visible in the former tumor bed (9–12 o’clock).
- DWI: High
signal (white arrows) is located contralaterally, outside the
original tumor bed.
This is caused by a susceptibility artifact from gas in the
rectal lumen.
Always correlate the location of high DWI signal with the original tumor bed.
This helps distinguish artifacts from true tumor-suspicious signal.
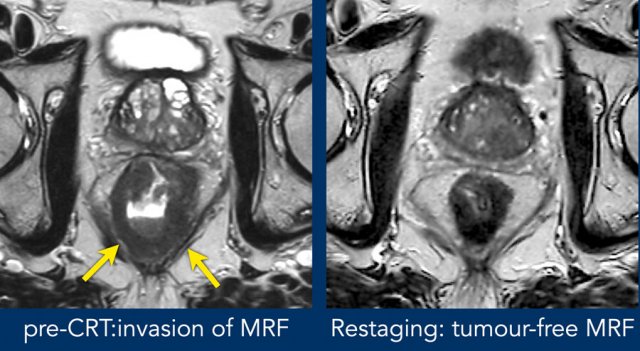
Post-CRT assessment of MRF involvement
Fat Plane Reappearance
When a fat plane reappears between the tumor bed and MRF after chemoradiotherapy (CRT), the risk of persistent MRF involvement is very low.
Diffuse Tumor Signal Infiltration
If diffuse MRF infiltration by intermediate tumor signal persists post-CRT, the risk of histopathologic MRF invasion is high (~90%).
Diffuse Fibrotic Infiltration
In cases with diffuse fibrotic MRF infiltration, the risk of MRF positivity at histopathology is ~50%.
Image Example
- Pre-CRT: Extensive MRF invasion from 4–8 o’clock (arrows).
- Post-CRT: Tumor has undergone fibrotic transformation and retracted from the MRF. A fat plane is now visible, with only minor fibrotic stranding—indicative of a tumor-free MRF at restaging (yMRF-).
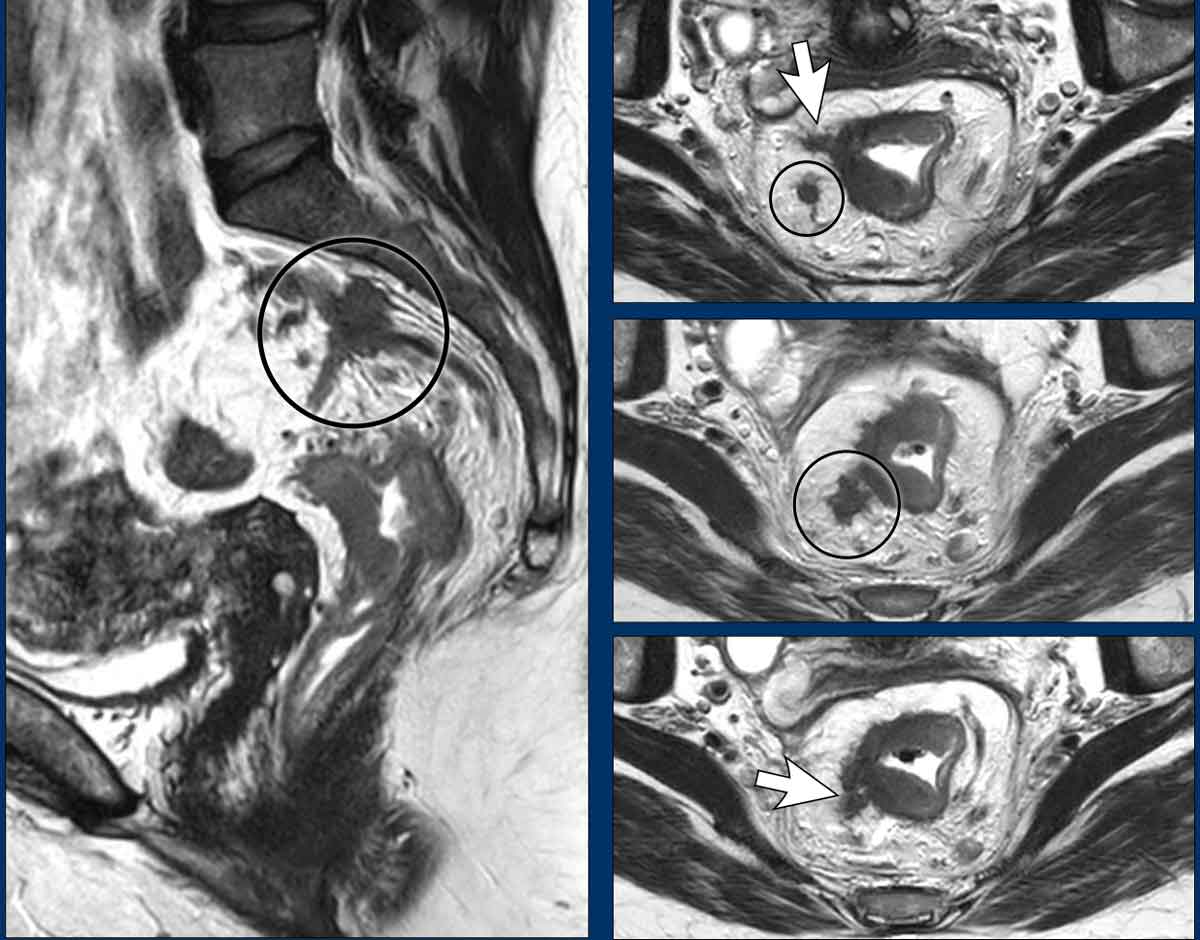
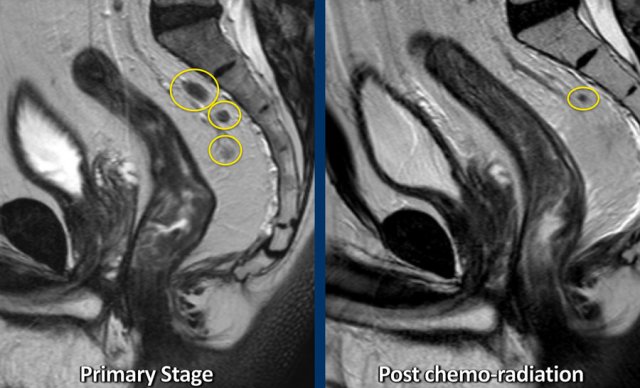 Example of a patient with several irregularly enlarged cN+ nodes at primary staging. After chemoradiotherapy, most nodes have disappeared and only a small node of < 5 mm remains, indicative of a ycN0 stage.
Example of a patient with several irregularly enlarged cN+ nodes at primary staging. After chemoradiotherapy, most nodes have disappeared and only a small node of < 5 mm remains, indicative of a ycN0 stage.
Restaging lymph node status post-CRT (ycN-category)
MRI’s diagnostic performance for restaging lymph nodes after CRT is superior to primary staging. Post-CRT, most nodes shrink or disappear on MRI.
ESGAR Guidelines:
- ycN0: Nodes with short-axis diameter <5 mm on restaging MRI.
- ≥5 mm nodes: Assess risk for yN+ disease in the context of primary tumor response, as nodal response typically mirrors that of the primary tumor.
- ycN+: Clearly pathologic nodes (e.g., >9 mm).
Borderline Nodes (5–7 mm):
- Higher yN+ risk in patients with major residual tumor.
- Lower yN+ risk in patients with minor residual tumor or (near-)complete response.
- For organ-preservation candidates, consider the "test of time" (watch-and-wait): Stable or shrinking nodessuggest ycN0; growing nodes indicate yN+ disease.
Lateral Nodes:
- No validated restaging criteria or size cut-offs exist.
- Report size reduction of initially suspicious lateral nodes.
- Multidisciplinary assessment required to weigh persistent disease risk, considering size decrease, mesorectal node response, and primary tumor response.
- "Test of time" may be considered for ambiguous cases.
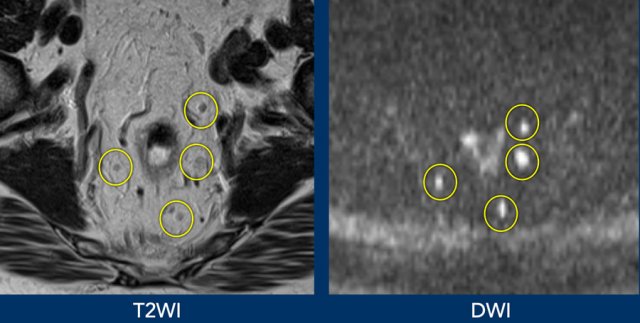
Lymph nodes, like all lymphoid tissues (e.g., spleen), have a dense cellular structure, resulting in restricted diffusion and high DWI signal.
DWI can enhance lymph node detection, especially in areas where nodes are easily overlooked, such as the internal iliac regions.
Limitation: DWI cannot characterize lymph nodes—both benign and metastatic nodes show high signal.
Image Example
- Improved lymph node visualization on DWI compared to T2-weighted imaging.
- DWI is not useful for assessing malignancy risk in these nodes.
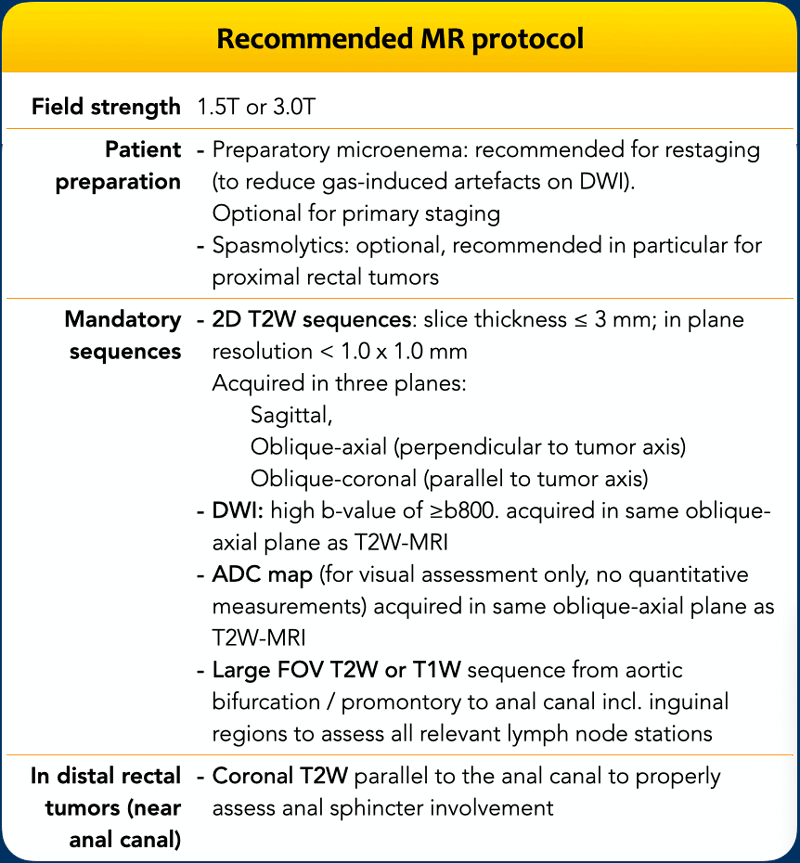
MR protocol
Hardware
- Field Strength: 1.5T or 3.0T
- Coils: Phased array external surface coils
- Endorectal Coil: Not recommended
Patient Preparation
- Spasmolytics: May be used to reduce bowel movement artifacts, especially for upper rectal tumors.
- Endorectal Filling: Not recommended—distension may interfere with accurate assessment of tumor distance to surrounding structures (e.g., mesorectal fascia).
- Gas Reduction: Self-administered micro-enema 15–30 minutes pre-MRI is recommended to minimize DWI susceptibility artifacts, particularly for restaging.
MR Series Planning
Sagittal Series
- Purpose: Tumor localization and planning of axial/coronal series.
- Field of View (FOV):
- Cranial border: Sacral promontory level
- Caudal border: Below the anal canal
Axial Series
- Orientation: Perpendicular to the tumor axis (oblique-axial)
- Purpose: Accurate assessment of tumor extension beyond the rectal wall and distance to the MRF
Coronal Series
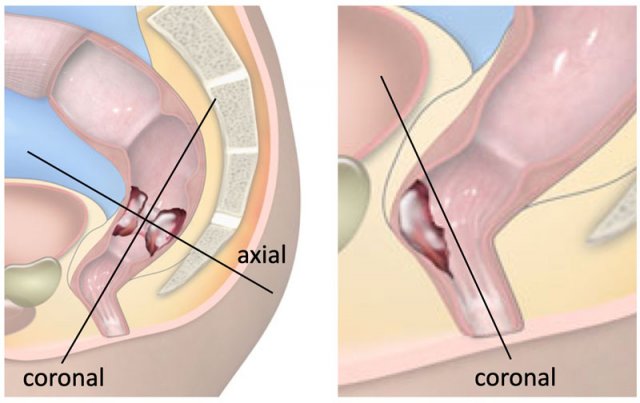
- Orientation: Parallel to the tumor axis (perpendicular to axial series)
- Special Consideration for Distal Tumors:
- If tumor is near/involving the anal canal, plan coronal sequences parallel to the anal canal or add an additional coronal plane parallel to the anal canal (right figure).
- Purpose: Optimal evaluation of anal sphincter invasion.
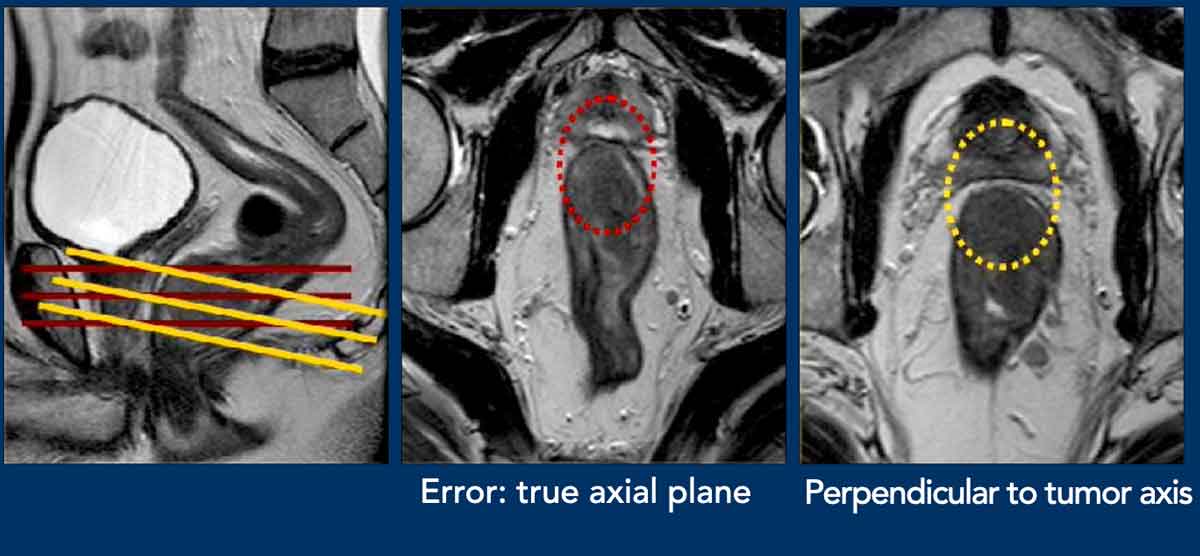
Pitfall – misinterpretation caused by incorrect angulation
Middle Image:
- Error: Axial view angled in the true axial plane, not perpendicular to the tumor axis of this distal rectal tumor.
- Result: False appearance of anterior MRF involvement (red circle).
Right Image:
- Oblique-axial view correctly angled perpendicular to the tumor axis.
- Result: Clear fat plane (>1 mm) visible between
tumor and MRF (yellow circle), confirming no MRF
involvement.
High Resolution 2D T2WI
A rectal MRI protocol
should routinely include high resolution 2D T2-weighted sequences in multiple
planes with a slice thickness of ≤3 mm and in plane resolution of < 1 x 1 mm.
As a rule of thumb, an in plane resolution of 0.6 x 0.6 mm results in good
quality, high-resolution images.
Although recent technical advances have improved the quality of 3D T2-weighted sequences, they are not yet commonly used as a replacement for 2D T2-weighted sequences.
DWI
DWI is recommended for all rectal MRI protocols.
It
can be useful for tumor and lymph node detection in primary staging and is
particularly useful for the detection of residual tumor within fibrosis after
neoadjuvant treatment.
There are some data suggesting that DWI can aid in detecting yEMVI and persistent yMRF involvement after CRT [ref1], [ref2], [ref3].
DWI Requirements:
- High b-value: ≥800 s/mm²
- ADC Maps: Must be generated and reviewed visually alongside DWI.
- Quantitative ADC: Not recommended for clinical (re)staging.
Image Example
- T2-weighted MRI: Tumor barely recognizable due to fecal obscuration.
- DWI: Tumor clearly detectable, demonstrating DWI’s advantage in challenging cases.
Field of View and additional sequences
Large FOV Pelvic Sequence
- Coverage: Full pelvis, from aortic bifurcation to distal anal canal margin, including inguinal regions and all relevant lymph node stations.
- Sequence Options: (Fast) T2-weighted or T1-weighted series.
- T1W Advantage: Useful for characterizing incidental findings (e.g., bone lesions, ovarian cysts).
Gadolinium-enhanced imaging
- Not required for standard staging —does not improve diagnostic accuracy.
T2-weighted
Fat-suppressed imaging:
- Not required for standard staging.
- T2-weighted fat-suppressed images: May be beneficial for patients with concomitant perianal fistulas or abscesses.
Surgical Procedures for Rectal Cancer
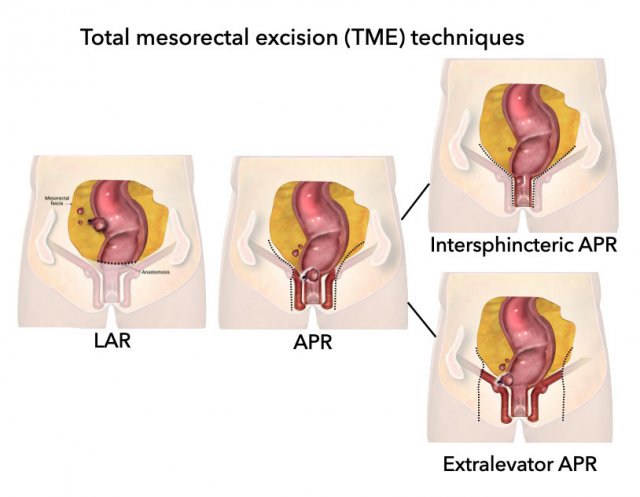
Total Mesorectal Excision (TME)
Is the standard procedure for rectal cancer and involves complete resection of the mesorectum along the mesorectal fascia, encompassing various techniques like LAR, APR, intersphincteric and extralevator APR.
Transanal TME (taTME) or ‘bottom up’ surgery combines
laparaoscopic surgery with a transanal approach.
Low Anterior Resection (LAR)
- Procedure: Anal canal preserved; anastomosis created between rectum and sigmoid colon.
- Indication: High rectal tumors with sufficient margin between tumor and anal canal.
- Anastomosis: Most commonly side-to-end.
Abdominoperineal Resection (APR)
- Procedure: En bloc resection of rectum and anal canal; permanent colostomy required.
- Indication: Low rectal tumors with close margin or anal canal involvement.
- Perineal Repair: Options include primary closure, omentoplasty, or plastic reconstruction (VRAM/ORAM myocutaneous flaps).
Intersphincteric APR
- Variation of APR where the external sphincter is spared.
Extralevator APR
- Extended procedure including levator ani muscles; indicated for tumors invading the pelvic floor (levator).
Beyond TME Surgery and Pelvic Exenteration
- "Beyond TME": Extended resections for tumors involving structures outside the mesorectal compartment.
- Pelvic Exenteration: Most extensive form; en bloc removal of rectum and one or more adjacent pelvic organs.
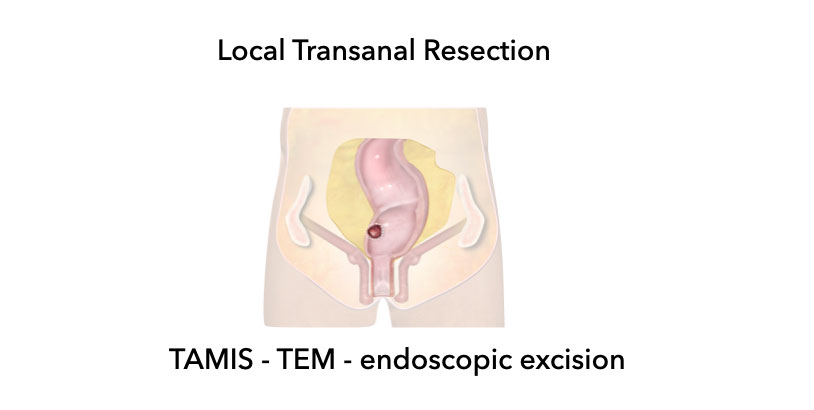
Local Transanal Resection
- Minimally invasive techniques for endoscopic excision of rectal tumors via the anus.
- Endoscopic Mucosal Resection (EMR) and Endoscopic Submucosal Dissection (ESD) are techniques for superficial excision for non-cancerous polyps and T1sm1/T1sm2 tumors.
- Transanal Minimally Invasive Surgery (TAMIS) and
Transanal Endoscopic Microsurgery (TEM) are full-thickness resection for T1 (and select small T2) tumors.
Organ-preservation
There is a growing shift toward minimally invasive or non-surgical approaches for rectal tumors showing complete or near-complete response after neoadjuvant treatment.
These strategies are now integrated into major clinical guidelines and include:
- "Watch-and-wait": Patients with a clinical complete response after neoadjuvant treatment defer surgery and undergo close surveillance.
- Local excision or radiotherapy: For patients with small tumor remnants.
Evolution of Organ-Preservation:
- Initially focused on "secondary organ-preservation"—patients receiving neoadjuvant chemoradiotherapy for tumor downsizing who achieved such a good response that surgery could be avoided.
- In clinical trials, (chemo)radiotherapy is selectively offered to certain lower-risk patients preferring organ preservation, with the primary intent of organ preservation from the start.
Treatment choices are multifactorial, balancing oncologic safety, patient preferences, and expected quality of life.
The success of these strategies depends on highly accurate post-treatment imaging.
MRI, combined with endoscopy and clinical examination, is central to both patient selection and surveillance during organ-preservation.
Video examples of Staging
Low Rectal cancer
In this case we demonstrate how to stage a low rectal cancer.
You can scroll through the images and then go to the video in wich we will explain the staging.
In this video we demonstrate how to stage a low rectal cancer.
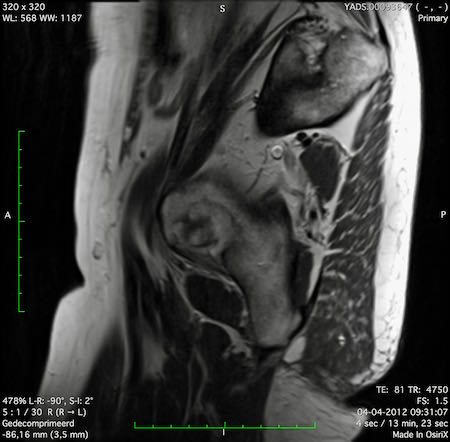
High Rectal cancer
In this case we demonstrate how to stage a high rectal cancer.
You can scroll through the images and then go to the video in wich we will explain the staging.
In this video we demonstrate how to stage a cancer in the upper or high rectum.
With special attention for the anterior peritoneal reflexion, mesorectal fascia and extramural vascular invasion.
If you want to scroll through the images yourself, you may first wanna look at the scroll images.
Charity
All profits from The Radiology Assistant are donated to Medical Action Myanmar, an organization led by Dr. Nini Tun and Professor Frank Smithuis Sr.—a professor at the University of Oxford and the brother of Robin Smithuis.
[Watch the Medical Action Myanmar video here.]
If you appreciate The Radiology Assistant, please consider supporting Medical Action Myanmar with a small donation.